Reclaiming Slick-Spots and Salty Soils
Soils, which do not respond to normal management practices, are called “problem soils.” Acid soils are often considered problem soils for this reason. Correction of acid soils by liming is discussed in OCES Fact Sheet PSS-2229. Two other kinds of problem soils are salty (saline) soils and slick-spot (alkali or sodic) soils. A third kind of problem soil often develops from slick-spots when they are poorly managed. This is the saline-alkali soil, formed when slick-spots become salty.
Although all problem soils may be identified with poor crop production, these soils have other similarities and differences, which are important to know before attempting to improve or reclaim them.
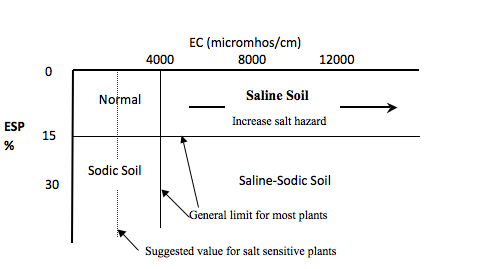
Saline soils are soils, containing at least 2,600 ppm soluble salts in the solution from a soil saturated with water. The salt content is estimated by laboratory measurement of how well the soil water conducts electricity, and saline soils are those with an electrical conductivity of 4,000 micromohs/cm (equivalent to 2,600 ppm soluble salts). This level of salts is high enough to reduce production of most crops.
Alkali soils are soils, which contain enough sodium (Na) to cause 15 percent of the cation exchange sites to be occupied by sodium. Sodium in the soil prevents clay particles (and other very small, colloidal sized particles such as humus) from coming together and forming large soil aggregates. When soil contains 15 percent or more exchangeable sodium most of the clay and humus particles are unattached or dispersed. These soils commonly have a pH of 8.5 or higher (alkali). The classification of salt affected soils is shown in Figure 1. Some Oklahoma soils become dispersed when the exchangeable sodium is as low as 7 percent. Productive agricultural soils often have less than 1 percent exchangeable sodium.
Figure 1. Classification of salt affected soils and plant growth response to EC and ESP.
Characteristics of Saline Soils
Small, Growing Areas Affected
Naturally developed saline soils usually represent only small areas of a field. Often these are low lying parts of the field, which may have poor internal soil drainage. Other small areas occur on slopes where erosion has exposed saline or alkali subsoil. Because low areas are frequently wet when the rest of the field is dry enough for cultivation, these small areas frequently are cultivated when the soil is too wet. This results in the soil becoming compacted in and around the area. Water does not move easily through the compacted soil so more water evaporates, leaving salts from the water to accumulate in the soil. As a result, the-affected area gets larger with time.
Poor Yield
Crop production is usually less than normal from salt affected areas. Yield reduction is greater in years of less than normal rainfall or when water stress has been a yield limiting factor in general. Salts “tie-up” much of the water in the soil and prevent plants from absorbing it. Seedlings are the most sensitive to water stress, and crop stand is reduced because of seedling death and poor yield results.
White Surface Crust
As water evaporates from saline soils, salts, which were in the water, are left behind to accumulate on the soil surface. The salts are light colored, and when accumulation has continued for several weeks, they form a very thin white film on the soil surface. During hot, dry weather the light film will show up first along edges of the salt problem areas. The center of these areas usually has the most salt and will dry out last.
Good Soil Tilth
Saline soils generally have excellent physical condition throughout the tillage depth. This is caused by the salts effectively neutralizing the negative charge of clay particles, allowing them to attach to one another. When these soils are not too wet, the soil is friable, mellow, and easy tilled. The appearance after tillage is that of a very productive soil.
High Soil Fertility
Soil which has been saline for several years will usually be very fertile, and high nitrogen (N), phosphorus (P), and potassium (K) soil test values are often a clue of a problem soil. These nutrients build up in salty areas when there is little nutrient removal by crops and the area is fertilized each year. Soil pH does not change with salt content and it can not be used as an indicator.
Characteristics of Alkali Soils
Except as noted, alkali soils have characteristics similar to saline soils. For this reason one problem soil may be confused for the other. Their differences, however, are important to note as they relate to correcting the problem soils.
Poor Soil Tilth
The excess sodium in alkali soils does not allow soil particles to easily attach to one another. As a result alkali soil is dispersed, and not friable and mellow like saline soil. Instead, alkali soil is greasy when wet (“slick-spot”), especially if it is fine textured, and often very hard when dry. This poor physical condition makes these soils difficult to manage. They are often either too wet or too dry for tillage. Poor seed germination and stand establishment are common because good seedbed preparation is seldom accomplished. As a result yields are usually lower than the rest of the field and fertility may build up.
Dark or Light Colored Surface
Soil colloids, which are floating in soil solution, are left as a thin film on the surface after water evaporates. The surface color will be darker than the rest of the field (black-alkali) when the particles are mainly humus and lighter (white-alkali) when the particles are mainly silt and salts. The salts show up as a film when the surface dries.
Droughty But Pond Water
Large pores or channels in the soil, which allow water entry and penetration, become plugged with dispersed clay and humus. As a result, the subsoil may be very dry even though water is ponded on the surface. Plants that do become established often suffer water stress and may eventually die from lack of water and/or oxygen.
Reclamation
In many instances saline soils and alkali soils can be reclaimed by following a definite series of management steps designed to leach or “wash out” the salts or sodium. The order and description of these steps follows.
Verify Problem
The first step to solving the problem is clearly identifying it. This is best done by having the soil tested. Suspected areas should be sampled separate from the rest of the field. It is best to sample during a dry period of the growing season when affected areas of the field can easily be identified by the poor crop growth. Samples should be taken at least one week from the last rain or irrigation and only the top three inches of soil should be sampled. Several small samples of the affected area should be combined in a plastic bucket and mixed to get a good sample.
About one pint of soil is required for the test, which is done by the OSU Soil Testing Laboratory. Samples should be submitted through local Extension office requesting a Salinity Management or Comprehensive Salinity test. Testing takes a 3 to 5 days and a small fee is charged to cover costs. This test will identify the type and seriousness of the problem.
Identify Cause
Whenever possible, it is important to find out what has caused the problem soil to develop. Knowing the cause can help in modifying the remaining reclamation practices and sometimes provide a clue as to how long it may take to complete the reclamation.
The four most common causes of saline and alkali soils in Oklahoma are: a) natural poor drainage; b) poor irrigation water; c) brine spills; d) exposure of saline or alkali subsoil due to erosion.
Poorly drained soils are simply soils which water does not easily penetrate. This condition may be a result of the soil having a high clay content, having a water table near the surface (within 10 feet), or existing in a low lying area of the field. In the later situation normally adequate internal drainage may not be able to handle runoff from the surrounding area. In some instances internal soil drainage is greatly reduced as a result of compacting the surface soil.
Use of poor quality irrigation water (see OSU Extension Facts PSS-2401) may cause problem soils to develop if special precautions are not taken. The problem develops most rapidly during extremely dry years when evaporation and the amount of irrigation are high. Internal soil drainage may be a contributing factor.
Problem soils sometimes develop “overnight” when brine solutions associated with oil and gas well activities spill onto the soil. Depending on the amount of brine solution spilled and the size of the area, the problem may be slight or very severe.
Whenever the source of salt or sodium causing the problem is external, such as runoff, seeps, irrigation water or spilled brine, it is important to eliminate that source as soon as possible.
Improve Internal Soil Drainage
There are no chemicals or soil amendments that can be added to the soil to “tie-up” or somehow inactivate soluble salts or sodium. Hence, the only way of lowering their concentration in the soil is to remove them. This can only be done by leaching (washing out) the salt or sodium downward out of the root zone. In order for this to happen internal drainage must be good so water can easily pass through the soil.
There are a number of ways internal drainage can be improved. Most are expensive, but when the problem is severe many will pay for themselves with time. Tile drains and open ditches are effective for removing subsoil water that accumulates due to a restrictive layer such as compacted clay or bed rock. Compacted soil layers near the surface can be broken up by subsoiling. This is effective only if done when the soil is dry enough to have a shattering effect and at best provides only temporary benefit.
Problem soils which have developed from use of poor irrigation water or brine spills may already have good internal soil drainage.
Add Organic Matter
Once internal drainage has been assured, the next important step is to improve water movement into the soil. Incorporating 10-15 tons per acre of organic matter into the top six inches of soil creates large pores or channels for water to enter. Even rainfall from intense storms is more effective because there is less runoff. In addition to improving water movement into the soil, the large pores lessen the capillary or wick-like upward water movement during dry periods. Any coarse organic material such as barn yard manure, straw, rotted hay, or crop residue is suitable. Make sure not to exceed N and P limits when using manure.
Add Gypsum to Slick-Spots
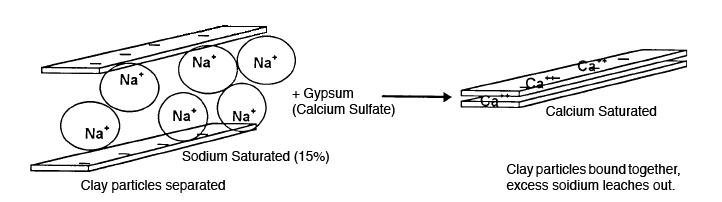
Up to this point the reclamation practices are the same for both saline and alkali soils. In either situation leaching is critical to remove salt or sodium. However, since high amounts of sodium absorbed to the soil are the cause of the alkali problem, the sodium must be loosened from the soil before it can be leached out. Gypsum is the most economical soil amendment for removing sodium from the soil particles. Gypsum is a slightly soluble salt of calcium and sulfate. This means that gypsum will react in the soil slowly, but for a long time. The reaction is illustrated in Figure 2.
The amount of gypsum required will vary widely depending upon the percentage of exchangeable sodium and the soil texture, as determined by the soil test. This relationship is shown in Table 1.
Figure 2. Gypsum applications are needed when the exchangeable sodium percentage (ESP) is greater than 15 percent. Calcium ions (Ca++) in gypsum replace sodium ions (Nat+) on the colloids, which results in improved soil conditions. See Table 1 for rate of gypsum to apply for certain conditions. Gypsum application does not benefit soils that are high in soluble salts. Such an application benefits only soils in exchangeable sodium as shown in Table 1.
Table 1. Gypsum Requirement in tons per acre as Related to Soil Texture and Sodium Percentage.
| Exchangeable Sodium Percentage | |||||
| Texture | 15 | 20 | 30 | 40 | 50 |
| Tons per acre | |||||
| Coarse | 2 | 3 | 5 | 7 | 9 |
| Medium | 3 | 5 | 8 | 11 | 14 |
| Fine | 4 | 6 | 10 | 14 | 18 |
When the required amount of gypsum exceeds five tons per acre, the rate should be split into two or more applications of no more than five tons at one time. Successive applications should not be made until time has allowed for some leaching to occur, and the need has been verified by a second soil test. The gypsum should be incorporated only to a depth of about one or two inches, enough to mix it well with the surface soil and keep it from blowing away.
Leach Soil
Leaching (or washing out) the soil is essential to reduce the amount of salts or sodium in the soil. In order for this leaching process to occur, water must enter the soil in excess of what is needed by growing crops and lost by evaporation. How fast and to what extent the reclamation is successful will depend on how much good quality water passes through the soil in a given period of time. The shorter the time interval over which excess water is applied, the more effective that amount of water is in reclamation. For this reason, rainfall is most effective when it falls on soil, which is already wet.
Avoid Deep Tillage and Establish Cover
Once the leaching process has been started, deep tillage such as moldboard plowing should be avoided for several years to promote uninterrupted downward movement of the salts. Such tillage tends to bring soil and salts from the depth of tillage up to the soil surface, and leaching must be started all over. As soon as the salt level in the soil will permit, a salt tolerant crop such as barley or bermudagrass should be established on the problem area to provide a cover for as much of each growing season as possible. It is especially important to have the cover crop during midsummer when evaporation is high. Adequately fertilized bermudagrass does a good job of drying the soil. To minimize soil compaction it should be cut for hay instead of pastured. Make sure to keep heavy equipment off the area when it is wet.
Some problem areas may be too salty to establish a cover crop until some salts have been leached out. When there is no longer a white salty film on the soil surface, following a week or two of dry weather, or when weeds begin to grow are clues indicating a cover crop can be established.
Wait
The final step in reclamation is simply to wait for the previous practices to work. Except for brine spills, these problem soils developed over a period of several years.
Reclamation may not take as long, but, depending on how well reclamation practices can be carried out, may take one or more years.
Alternative to Drainage – Reclamation
Learn to Live With It
The key to successful reclamation is good internal soil drainage. If salts or sodium cannot be leached out, the soil cannot be reclaimed. However, most soils have some internal soil drainage, and although drainage may not be good, over several years time it may be sufficient to lower the salt concentration to near normal. During this time it will be important to practice some of the same steps outlined above. Especially important are the following:
- Avoid excessive fertilization.
- Avoid traffic on field when wet.
- Apply gypsum to slick spots.
- Establish cover crop.
- Maintain high level of crop residue.
- Be patient!
Depending on the severity of the problem it may be necessary to select a different crop than what has been grown in the past. A list of crops and their tolerance to salt is provided in Table 2.
Table 2. The Relative Salt Tolerance of Crops* (In Decreasing Order of Tolerance).
| Tolerant | Moderately | Sensitive |
|---|---|---|
| Field Crops | ||
| 10, 400 ppm | 7,800 ppm | 2,600 ppm |
| Barley (grain) | Rye (grain) | Field Beans |
| Sugar Beet | Wheat (grain) | |
| Cotton | Oats (grain) | |
| Grain sorghum | ||
| Soybeans | ||
| Corn | ||
| Sunflower | ||
| 7,800 ppm | 3,900 ppm | |
| Forages | ||
| 11,700 | 7,800 ppm | 2,000 ppm |
| Alkali sacaton | White sweetclover | Peanuts |
| Saltgrass | Yellow sweetclover | White Dutch Clover |
| Bermudagrass | Perennial ryegrass | Red clover |
| Rhodesgrass | Dallisgrass | Ladino clover |
| Rescue grass | Sudangrass | |
| Barely (hay) | Alfalfa | |
| Birdsfoot trefoil | Rye (hay) | |
| Wheatgrass | Wheat (hay) | |
| Oats (hay) | ||
| Blue grama | ||
| Fescue | ||
| Smooth bromegrass | ||
| 7,800 ppm | 2,600 ppm | 1,300 ppm |
| Vegetable Crops | ||
| 7,800 ppm | 6,500 ppm | 2,600 pm |
| Garden beets | Tomato | Radish |
| Kale | Broccoli | Celery |
| Asparagus | Cabbage | Green beans |
| Spinach | Cauliflower | |
| Lettuce | ||
| Sweet corn | ||
| Potatoes | ||
| Sweet potatoes and yams | ||
| Bell pepper | ||
| Carrot | ||
| Onion | ||
| Peas | ||
| Squash | ||
| Cucumber | ||
| 6,500 ppm | 2,600 ppm | 1,950 ppm |
| Fruit crops | ||
| High Salt Tolerance | Medium Salt Tolerance | Low Salt Tolerance |
| Grape | Pear | |
| Cantaloupe | Apple | |
| Plum | ||
| Apricot | ||
| Peach | ||
| Strawberry |
* Salt tolerance values at which 50% yield reduction may be expected compared to nonsaline conditions.
Summary
The following major points are discussed in this Fact Sheet:
- Saline soils and slick-spots soils have many similar, characteristics, but differences must be recognized for proper reclamation.
- Problem soils can be improved.
- Reclamation may be expensive and require major changes in soil and crop management.
- Reclamation may take a long time.
Hailin Zhang
Director, Soil, Water and Forage Analytical Laboratory