Smallseed Falseflax Management in Winter Wheat
Smallseed Falseflax Identification and Biology
A native to Europe, smallseed falseflax (Camelina microcarpa Andrz. Ex DC.) was first introduced to North America in the 19th century, likely as a contaminant in flax seed (Linum usitatissimum L.) and other crops (Francis and Warwick 2009). Since its introduction, it has become a common weed found in agricultural crops but has recently been considered as a potential oil seed crop. As a pest, it is most commonly found in the southern Great Plains in cool-season crops such as winter wheat. Although falseflax has not been noted by producers in Oklahoma to be of high economic importance, it is still an undesired species competing on a wide geographical area.
Figure 1. More lobed horseweed leaves (A)
Falseflax can appear similar to horseweed (Conyza canadensis L.), a weed that has a considerable impact on agriculture (SeeFact Sheet PSS-2793, Horseweed Management in Oklahoma Winter Wheat). Much like horseweed, falseflax is a winter annual that develops a basal rosette covered in dense hairs (Figure 1). However, the rosette leaves are not lobed like horseweed. As falseflax matures, it develops an erect stem that is either simple or branched and can reach one meter in height. Like many species in the Brassicaceae family, it has a raceme inflorescence with a terminal cluster of small, four-petaled, pale yellow flowers. Once pollinated, these flowers develop into small, pear-like siliques or “pods” (Figure 2). Falseflax is capable of producing almost 13,000 seeds per plant (OA Stevens 1957). These seeds could result in potential dockage issues at the grain elevator
Smallseed Falseflax Chemical Management
Other potential reasons for concern of falseflax presence outside of crop competition include herbicide resistance and potential out-crossing with other mustard species.
Less lobed, hairier smallseed falseflax leaves (B).
Figure 2. Close-up of smallseed falseflax pear-like reproductive
structure.
Through a whole plant dose-response study, Hanson et al. (2004) confirmed acetolactate synthase (ALS) resistance to metsulfuron (Ally® XP) and chlorsulfuron (Glean® XP) occurring naturally in a falseflax population in Oregon. This was the result of a single point mutation within falseflax that, in other studies, resulted in resistance to four of the five chemical groups that make up the ALS site of action (Hanson et al. 2004; Tranel and Wright 2002). Use of ALS herbicides in small grain producing regions is high and therefore the continued selection of herbicide resistant biotypes is of great concern.
Management of falseflax in grain-producing cereal crops can be accomplished in several
ways. Control of many broadleaf weeds is achieved most commonly by the use of either
ALS inhibiting herbicides, synthetic auxin herbicides or mixtures of the two sites
of action; however, few studies have evaluated falseflax response to various herbicides.
According to an OSU Extension fact sheet (PSS-2787, Weedy Mustards
of Oklahoma), Ally®, Beyond® (in CL and CL+ Wheat), Olympus®, Outrider®, PowerFlex® HL and premixes of Quelex®, Finesse® Cereal & Fallow
and Sentrallas® are all effective at controlling falseflax that is not ALS resistant.
Other herbicide options include synthetic auxin herbicides like 2,4-D, dicamba or
4-Hydroxyphenylpyruvate dioxygenase (HPPD)/ Photosystem II (PS II) premixes like Huskie®
or Talinor®. One trial
was conducted at the OSU North Central Research Station during the 2016-17, 2017-18
and 2019-20 winter wheat growing seasons. The goal of the study was to evaluate smallseed
falseflax control with previously available products as well as newer products. The
herbicides and rates used are included in Table 1.
Field Research Findings and Recommendation
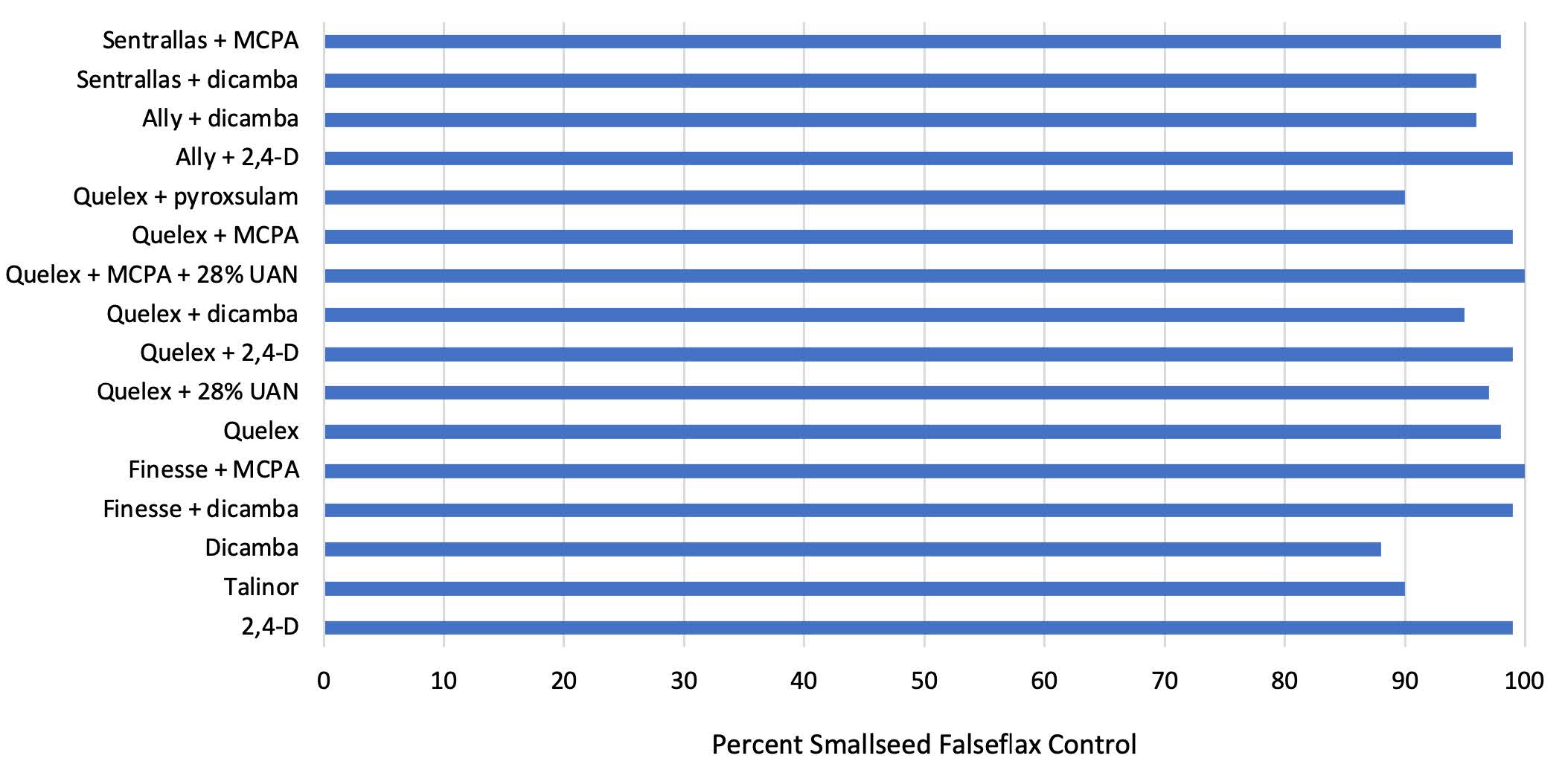
Averaged across years, falseflax control was 88% or greater for all treatments with
no statistical separation (Figure 3). Numerically, all treatments achieved 95% control
or
greater except Talinor® and Quelex + Powerflex® HL, which both provided 90% control
of falseflax. Control with dicamba was at 88%. Herbicide treatment also did not affect
grain yield. A major benefit from the relatively high level of falseflax control provided
by all herbicide treatments is that producers battling falseflax have several options.
They also have options with a relatively wide range in price as dicamba or 2,4-D alone
can cost less than $2.00 per acre for the herbicide. The high efficacy of the treatments
tested allows winter wheat producers the opportunity to rotate through the use of
multiple herbicide sites of action to control falseflax, thus reducing the potential
to select for herbicide resistance in this species. Additionally, due to the high
efficacy of all treatments containing an ALS herbicide, herbicide resistance is not
suspected in this
population contrary to what Hanson et al. (2009) found in the previously mentioned
population in Oregon.
| Herbicide common names | Brand names or designations | Application rates (per acre) |
|---|---|---|
| 2,4-D Ester | 2,4-D LV 6 | 5.7 fl oz (in tank-mix) 10.7 fl oz (alone) |
| Bicyclopyrone + bromoxynil | Talinor® | 18.2 fl oz |
| Dicamba | Banvel® | 2 fl oz (in tank-mix with Ally® XP) 4 fl oz (alone, in tank-mix with Finesse® or Sentrallas®) |
| Halauxifen + florasulam | Quelex® | 0.75 oz |
| MCPA Ester | MCPA Ester 4 | 10.8 fl oz (in tank-mix with Quelex®) 17.3 fl oz (in tank-mix with Finesse®) |
| Metsulfuron | Ally® XP | 0.1 oz |
| Chlorsulfuron + metsulfuron | Finesse® Cereal & Fallow | 0.4 oz |
| Pyroxsulam | PowerFlex® HL | 2 oz |
| Thifensulfuron + fluroxypyr | Sentrallas® | 10 fl oz |
Figure 3. End-of-season smallseed falseflax control following applications of 1- to 3-inch rosettes at the OSU North Central Research Station during the 2016-17, 2017-18 and 2019-20 winter wheat growing seasons. All treatments that included an ALS herbicide were applied in tank-mix with NIS at 0.25% vol/vol. Water was used as the sole carrier except where ‘28% UAN’ is noted.
References
Francis A, Warwick SI (2009) The biology of Canadian weeds. 142. Camelina alyssum (Mill.) Thell.; C. microcarpa Andrz. ex DC.; C. sativa (L.) Crantz. Can. J Plant Sci 791-810
Hanson BD, Park KW, Mallory-Smith CA, Thill DC (2003) Resistance of Camelina microcarpa
to acetolactate synthase inhibiting herbicides. Weed Res 44:187-194
Stevens OA (1957) Weights of seeds and numbers per plant. Weeds 5:46-55
Tranel PJ, Wright TR (2002) Resistance of weeds to ALSinhibiting herbicides: what
have we learned? Weed Sci 50:700-712