Nitrous Oxide Emissions from Soil
Introduction
Nitrous oxide (N2O) is now the number one ozone depleting gas emitted by human activities. In addition, it is listed as one of the top three greenhouse gases (GHG) of concern by the EPA. Therefore, N2O emissions have become an important factor to consider for agricultural production in light of recent discussions regarding climate change. Although the N2O concentration in the atmosphere is generally very low (approximately 0.32 ppm) compared to that of carbon dioxide (CO2) (approximately 380 ppm), it is a very potent greenhouse gas with 310 times the insulating effect of CO2 and persists in the atmosphere for 120 years (USEPA 2012).
Soil management in agricultural systems is responsible for 69 percent of the N2O emissions in the U.S. This makes up 3 percent of the total emissions of all GHG’s in the U.S. (USEPA 2012). Nitrous oxide emissions are of the result of natural processes occurring in the soil. Many factors influence the emission of N2O such as soil moisture, temperature, microbial activity, aeration and organic matter content. Primarily, N2O is produced during the microbial process of denitrification, in which nitrate (NO3–) is converted to N2 gas. When NO3– is not completely converted to the harmless N2 gas, the resulting byproduct is N2O. Denitrification occurs under conditions of limited oxygen availability in the soil environment. To a lesser extent, N2O can also be produced during nitrification, which again is a microbial process whereby ammonium (NH4+) is converted to NO3–. This reaction can occur anytime under aerobic conditions where NH4+ concentrations, soil moisture and temperature are adequate. When the concentration of inorganic nitrogen (NO3– and NH4+) is increased through applying commercial fertilizers or mineralization of organic N sources (manure or cover cropped legumes), N2O emissions are increased above ambient levels. However, N loss as N2O is not as great as N loss due to leaching or volatilization and is unlikely to impact crop growth. Due to concerns about its environmental impact, its management is becoming increasingly important.
Managing N2O Emissions from Soil
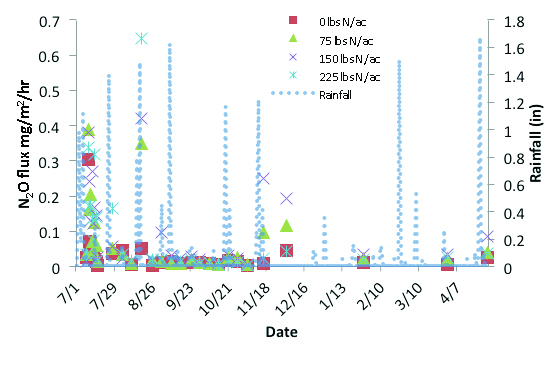
Nitrous oxide cannot be sequestered like CO2 in biomass or soil. Therefore, the only way to reduce atmospheric concentrations of N2O is to reduce its emissions. Emissions can effectively be reduced by reducing or minimizing the concentration of inorganic N in soils, especially during periods when denitrification or nitrification are most likely to occur (i.e. high temperatures and high soil moisture). Emissions of N2O are stimulated by rainfall (Figure 1). However, not all rainfall events will produce emissions; there must be enough rain to fill a soil’s pore space to 60 percent water. When this happens, the soil moisture is sufficient to stimulate microbial activity and deplete the oxygen content of the soil system, thereby producing N2O gas. The magnitude of these emission events is dictated by the concentration of inorganic N in the soil, the rate at which soil moisture drops below the threshold of 60 percent water filled pore space, and soil temperature. Therefore, soil type, timing of N fetilizer application, crop growth stage and crop type (water and N use by crop) will all impact N2O emissions. The most effective means of reducing N2O emissions are to: minimize N application rates; apply fertilizers during periods of rapid crop growth (i.e. when crop demands are high, which results in rapid inorganic N uptake, and/or when soil moisture and temperature are low; or utilize slow release fertilizers that minimize the concentration of inorganic N in the soil system.
Figure 1. Large rainfall events stimulated soil N2O emissions in a forage sorghum field during the 2010 crop year.
Enhanced Efficiency Fertilizers
The use of enhanced efficiency fertilizers such as those containing urease inhibitors, nitrification inhibitors and polymer coatings to reduce N2O emissions has received a great deal of attention in recent years. It was assumed because these products reduce the rate of inorganic N release to the soil environment, they would decrease N2O emissions. However, the results have shown this is not the case for all enhanced efficiency fertilizers. In fact, a recent literature review (Akiyama et al. 2009) on the subject found urease inhibitors are not effective at reducing N2O emissions. In contrast, nitrification inhibitors were found to significantly reduce N2O. Polymer coated fertilizers did reduce N2O emissions, but only in specific soil environments. Polymer coated fertilizers were only effective on poorly drained soils and provided little benefit on well drained soils.
Split N applications
One simple management practice that can increase N use efficiency (NUE) and reduce N2O emission is split N fertilizer applications. This practice is effective because it reduces the total fertilizer N required to optimize crop yield, and it decreases the amount of N applied at or near planting when crop uptake is low. These two factors decrease the concentration of N found in the soil system during the growing season. An additional benefit of this practice in a winter wheat system is that much of the N is applied in the winter instead of applying all of it when temperatures are warm before or at planting.
Cover Crops
Utilization of cover crops has also been suggested as a viable practice to reduce N2O emissions. Two options for use of cover crops to reduce N2O emissions are promising. First, planting non-legume cover crops that scavenge N from the soil system during fallow periods can reduce N2O emissions from residual N left over after the cash crop is harvested. Upon termination, the slow release of N back to the soil system can decrease fertilizer N requirements and the overall inorganic N concentration of the soil system. A second and perhaps more obvious option is the use of leguminous cover crops. After termination, leguminous cover crops can slowly release N to the cash crop, thereby reducing fertilizer N requirements.
Variable Rate N fertilizer Management
Perhaps the best way to reduce emissions is using variable rate N management in combination with cover crops and split applications. There are various management options to achieve variable rate N applications. An option proven in Oklahoma is the sensor-based N management. In fact, the use of sensor-based N management has been shown to increase NUE by more than 15 percent (Raun et al. 2002), meaning more N is used in the crop and less is available to environmental losses such as N2O emissions. This practice uses N-Rich strips to provide an in-season estimate of yield and a nitrogen response index. From these, a site specific N requirement is determined. This technology can be used to provide an optimum N rate on a whole field basis or to vary the rate of N application within the field. Increasing the resolution will increase the benefit, but also increases the cost of this management option. This technology allows producers to adapt to the growing conditions in each field every year. Without the use of this type of technology, the producer is left to guess the appropriate rate or simply apply a fixed rate regardless of the yield potential for that particular season. In the interest of decreasing N2O emissions, it is easy to suggest that the fixed rate of N simply be reduced. However, this could result in a decrease in the long-term average yields because the crop would be left deficient of N in high yielding years and still receive excess N during poor yielding years.
Nitrogen-rich strips and sensor-based N management are particularly useful for managing N recovery from cover crops. The rate of N release from decomposing cover crops can be inconsistent; therefore it is difficult to know how much N they will provide to the following cash crop. Nitrogen-rich strips and sensor based N management can reduce this uncertainty.
Oklahoma State University Extension publication E-1022, Nitrogen Rich Strips, provides more information on nitrogen-rich strips and sensor based nitrogen management practices. The local county Extension office also can provide additional information as well as a sensor, if you are interested in trying this technology.
Summary
Nitrous oxide emission from soils is a developing environmental issue that Oklahoma agriculture will face as concerns about climate change and ozone depletion continue to develop. Fortunately, it appears that some of the best management practices developed to increase nitrogen use efficiency and decrease water pollution will be effective at reducing N2O emissions. To prevent yield losses, while decreasing N2O emissions, those practices that optimize NUE while minimizing inorganic N concentrations in the soil during periods of warm wet conditions will be most effective.
References
Akiyama, H., X. Yan, and K. Yagi. 2009. Evaluation of effectiveness of enhanced-efficiency fertilizers as mitigation options for N2O and NO emissions from agricultural soils: meta-analysis. Global Change Biology 16(6): 1837-1846.
Raun, W. R., J. B. Solie, G. V. Johnson, M. L. Stone, R. W. Mullen, K. W. Freeman, W. E. Thomason and E. V. Lukina, 2002: Improving Nitrogen Use Efficiency in Cereal Grain Production with Optical Sensing and Variable Rate Application. Agron. J. 94, 815-820.
USEPA, 2012. Inventory of U.S. Greenhouse Gas Emissions and Sinks: 1990-2010. In: U. S. E. P. Agency ed. Washington, DC.
Tracy M. Wilson
Graduate Research Assistant
Jason Warren
Soil and Water Conservation and Management Extension Specialist
Brian Arnall
Precision Nutrient Management Extension Specialist