Implants and Their Use in Beef Cattle Production
The term “implant” is used to refer to a group of products used in the cattle industry that increase the rate and efficiency of growth, both metabolic and economic. Implants contain natural or synthetic compounds that produce physiological responses in the animal similar to natural hormones. Implants are typically made of a powder that is compressed into a small pellet. The pellet is placed, or implanted, under the skin on the backside of the animal’s ear. Each type or brand of implant has a specific applicator, referred to as an implant gun, which is used to properly administer the implant.
Types of Implants
There are three types of compounds used in implants: estrogens, androgens and progestins. Estrogens mimic the effects of the naturally occurring hormone estrogen. Estradiol benzoate, estradiol 17-beta and zeranol are the primary estrogenic compounds used in implants. Alternatively, androgenic compounds mimic the effects of the naturally occurring hormone testosterone. Testosterone propionate and trenbolone acetate (TBA) are the principal androgenic compounds used in implants. Synthetic progesterone also is used in implants; however, its effect on the animal is less pronounced than the other two hormone analogues. Table 2 (suckling calves), Table 3 (stocker cattle) and Table 4 (finishing) list compound combinations and dosages supplied in commercially available implant products. Some commercially available implants have singular hormone activity, such as Ralgro®, Encore® and Compudose® with only the estrogenic analogues and Finaplex®-H has only testosterone-like activity (TBA) but most have combinations of hormone analogues.
Safety of Implants
There is much concern expressed by consumer groups in the news and social media about using growth promoting hormones in beef production systems. Table 1 shows the estrogenic activity of foods commonly consumed in the U.S. Beef from steers and heifers fed for slaughter have a very low level of estrogenic activity, regardless of implant status. In fact, ice cream contains 272 times more estrogen than implanted beef (Preston, 1997). Common grain and vegetable sourced foods such as peas, wheat germ and cabbage have 180 to 1,000 times the estrogenic activity of implanted beef. Natural estrogen production in humans is much higher than many expect; a pregnant female produces 90,000,000 nanograms of estrogen/day, a non-pregnant adult female 5,000,000 nanograms of estrogen/day, an adult male 100,000 nanograms of estrogen/day and a pre-pubertal child 40,000 nanograms of estrogen/day (Preston, 1997). So, the safety of growth promoting implants is certain. The safety of implants is assured when FDA-approved products are used according to their labels. History and several organizations including, but not limited to, the U.S. FDA, the World Health Organization and the Food and Agriculture Organization have concluded the use of implants in beef production poses no safety risk to consumers.
Table 1. Estrogenic activity per 4-ounce serving of several common foods*
| Food | Estrogen, ng |
|---|---|
| Soybean oil | 226,757 |
| Cabbage | 2,721 |
| Wheat germ | 453 |
| Peas | 453 |
| Eggs | 3,968 |
| Ice Cream | 680 |
| Milk | 15 |
| Beef from pregnant cow | 159 |
| Beef from implanted cattle | 2.5 |
| Beef from non-implanted cattle | 1.8 |
a adapted from Preston (1997).
Current Use of Implants
Implants have a long history of use in the beef cattle industry. The first commercial implant was introduced in 1957. Since then, the use of implants has been widely adopted by the cattle feeding and stocker sectors of the beef industry. According to the 2011 USDA NAHMS Feedlot Survey (USDA NAHMS, 2013), up to 94% of steers and heifers are implanted at least one time during the finishing phase.
In the Southern Great Plains region (Kansas, Oklahoma and Texas) a survey of stocker cattle operations indicted 77% of stocker calves are implanted (Asem-Hiablie et al., 2015), but use of implants is much lower in cow-calf operations. In Oklahoma, 58.6% of stocker cattle operations always implanted their steers, while only 28.8% of stocker cattle from stocker operations that included a cow-calf component were implanted (Johnson et al. 2008). A recent Oklahoma survey of 729 producers who received the Beef Cattle Manual (Vestal et al., 2007) showed 37% of cow-calf producers with larger operations (more than 100 cows) indicated they implanted their steer calves, while only 9% of cow-calf producers with smaller operations (fewer than 100 cows) implanted their steer calves.
Effect of Implants on Beef Cattle Performance
Nursing Calves
Implant products are available for calves weighing less than 400 pounds (Table 2). Implants approved and labeled for use in nursing calves generally contain a lower dose of the active ingredient compared to products cleared for use in older cattle (Tables 3 and 4). These implants are typically administered when the calves are between 2 months and 4 months of age. Research has shown that implants given during the suckling phase will increase average daily gain (ADG) of steer calves by approximately 0.10 pound per day. The response in heifers is slightly lower. Zeranol and estradiol benzoate/progesterone implants appear to produce a slightly better response than estradiol 17-beta products.
Most calf implants are designed for payout in approximately 100 days to 120 days. Calves should be 30 days (Ralgro®) to 45 days (Synovex®-C or Component® EC w/ Tylan®) old before they are implanted. Refer to manufactures label for approved timing. Bull calves intended for breeding should not be implanted. Bull calves not intended for breeding should be castrated at the time of implanting, as one effect of the implant is possible inhibited scrotal development, which makes later castration more difficult.
Table 2. Beef cattle implants approved for use in sucking calves and calves less than 400 pounds.
| Steer | Heifer | Implant | Company | Ingredient/dose | Relative Potency | Payout |
|---|---|---|---|---|---|---|
| x | x | Ralgro® | Merck | 36 g zeranol | Low | 70 to 100 |
| x | Compudose® | Elanco | 25.7 mg estradiol | Moderate | 200 | |
| x | Encore® | Elanco | 43.9 mg estradiol | Moderate | 400 | |
| x | x | Synovex®-C | Zoetis |
100 mg progesterone
10 mg estradiol benzoate |
Low | 70 to 100 |
| x | x | Component® E-C w/ Tylan® | Elanco |
100 mg progesterone
|
Low | 70 to 100 |
1 Local antibiotic.
Table 3. Beef cattle implants approved for use in stocker calves and growing calves in confinement more than 400 pounds.
| Steer | Heiffer | Implant | Company | Ingredient/dose | Relative Potency | Payout |
|---|---|---|---|---|---|---|
| x | x | Ralgro® | Merck | 36 g zeranol | Low | 70 to 100 |
| x | Compudose® | Elanco | 25.7 mg estradiol | Low | 200 | |
| x | Encore® | Elanco | 43.9 mg estradiol | Low | 400 | |
| x | Synovex®-S | Zoetis |
200 mg progesterone
|
Moderate | 90-120 | |
| x | Component® E-S w/ Tylan® | Elanco |
200 mg progesterone
|
Moderate | 90-120 | |
| x | Synovex®-H | Zoetis |
200 mg testosterone propionate
|
Moderate | 90-120 | |
| x | Component® E-H w/ Tylan® | Elanco |
200 mg testosterone propionate
|
Moderate | 90-120 | |
| x | x | Revalor®-G | Merck |
40 mg trenbolone acetate
|
Moderate | 100-140 |
| x | x | Component® TE-G w/ Tylan® | Elanco |
40 mg trenbolone acetate
|
Moderate | 100-140 |
| x | x | Synovex® One Grass | Zoetis |
150 mg trenbolone acetate
|
Moderate | 180-200 |
1 Local antibiotic.
Table 4. Beef cattle implants approved for use in finishing cattle in confinement more than 400 pounds.
| Steer | Heifer | Implant | Company | Ingredient/dose | Relative Potency | Payout |
|---|---|---|---|---|---|---|
| x | x | Ralgro® | Merck | 36 g zeranol | Low | 70 to 100 |
| x | x | Compudose® | Elanco | 25.7 mg estradiol | Low | 200 |
| x | x | Encore® | Elanco | 43.9 mg estradiol | Low | 400 |
| x | Synovex®-C | Zoetis |
100 mg progesterone
10 mg estradiol benzoate |
Low | 70 | |
| x | Synovex®-S | Zoetis |
200 mg progesterone
|
Moderate Low | 90 to 120 | |
| x | Component® E-S w/ Tylan® | Elanco |
200 mg progesterone
|
Moderate Low | 90 to 120 | |
| x | Synovex®-H | Zoetis |
200 mg testosterone propionate
|
Moderate Low | 90 to 120 | |
| x | Component® E-H w/ Tylan® | Elanco |
200 mg testosterone propionate
|
Moderate Low | 90 to 120 | |
| x | x | Synovex® Choice | Zoetis |
100 mg trenbolone acetate
|
Moderate High | 100 to 140 |
| x | Revalor®-IS | Merck |
80 mg trenbolone acetate
|
Moderate High | 100 to 140 | |
| x | Component® TE-IS w/ Tylan® | Elanco |
80 mg trenbolone acetate
|
Moderate High | 100 to 140 | |
| x | Revalor-®IH | Merck |
80 mg trenbolone acetate
|
Moderate High | 100 to 140 | |
| x | Component® TE-IH w/ Tylan® | Elanco |
80 mg trenbolone acetate
|
Moderate High | 100 to 140 | |
| x | Revalor®-S | Merck |
120 mg trenbolone acetate
|
High | 100 to 140 | |
| x | Component® TE-S w/ Tylan® | Elanco |
120 mg trenbolone acetate
|
High | 100 to 140 | |
| x | Revalor®-H | Merck |
140 mg trenbolone acetate
24 mg estradiol benzoate |
Hight | 100 to 140 | |
| x | Component® TE-H w/ Tylan® | Elanco |
140 mg trenbolone acetate
14 mg estradiol benzoate
29 mg tylosin tartrate1 |
High | 100 to 140 | |
| x | x | Revalor® 200 | Merck |
200 mg trenbolone acetate
|
High | 100 to 140 |
| x | x | Component® TE-200 w/ Tylan® | Elanco |
200 mg trenbolone acetate
|
High | 100 to 140 |
| x | Revalor®-XS | Merck |
200 mg trenbolone acetate
|
High | 200 | |
| x | Revalor®-XH | Merck |
200 mg trenbolone acetate
|
High | 200 | |
| x | Finaplix®-H | Merck | 200 mg trenbolone acetate | High | 70 to 100 | |
| x | x | Synovex® Plus | Zoetis |
200 mg trenbolone acetate
|
High | 150 to 200 |
| x | x | Synovex® One Feedlot | Zoetis |
200 mg trenbolone acetate
|
High | 200 |
1 Local antibiotic
Nursing Bull Calves versus Nursing Implanted Steer Calves
Many producers follow the practice of leaving bull calves intact until weaning rather than castrating them. The idea is that natural hormones produced in the testicles increase ADG and weaning weight of the calves. Numerous research trials have shown that implanted steer calves gain at a rate equal to, or greater than, bull calves. Castrating bulls as small calves, as opposed to when they are older, reduces overall stress on the calf. The stress and hormonal effects of castration at weaning can reduce post-weaning gain potential and the calf’s ability to withstand diseases typically associated with weaning and marketing. This difference in post-weaning performance of bulls versus steers is recognized by cattle buyers. This is indicated by the fact that steers will command a $5 to $10 per cwt premium over intact bull calves. Producers wanting to maximize the value of male calves at weaning should consider early castration at birth or at two months to four months of age and use an implant approved for nursing calves.
Implanting Replacement Heifers
Producers often raise the question, “Is it safe to implant replacement heifers?” Research has shown heifer calves intended for use as breeding animals can be implanted one time between 45 days of age and weaning with no significant effect on subsequent conception rates or calving difficulty. Heifers implanted immediately at birth, following weaning or multiple times prior to weaning had significantly lower conception rates compared to heifers receiving a single implant prior to weaning.
Most producers should be able to identify potential replacements heifers at weaning. The producer then can implant the stocker heifers to improve gain and not implant the heifers intended for breeding.
Research has clearly revealed there is little, if any, detrimental effects of administering
growth-promoting implants to replacement heifers at the time of branding (2 months
to 4 months of age) or at the time of weaning. In fact, in research trials where one
implant was administered to heifer calves between 30 days of age and weaning, calving
difficulty was not influenced and fertility was only slightly reduced: a 1% to 3%
reduction in pregnancy rate (Selk, 1997). In two recent studies (Rosasco et al., 2018
and 2019), implants administered at branding time (3 months of age) or at weaning
did not influence subsequent reproductive performance of retained females. On the
other hand, weight gain is consistently improved when heifers are implanted at branding
or at weaning (Selk, 1997; Rosasco et al., 2018 and 2019).
Additional research has shown heifers implanted at birth and close to puberty (generally
around 9 months to 14 months of age) had substantially reduced fertility (7% to 39%
reduction in pregnancy rate; Selk, 1997) compared to nonimplanted heifers. Similarly,
heifers implanted more than once had substantially reduced fertility. Therefore, heifers
potentially kept as replacement females should either not be implanted at all, or
they should be implanted only one time between 30 days of age and weaning. Replacement
heifers should not be implanted prior to 30 days of age or after about 7 months of
age, and they should never be implanted more than once.
Stocker Calves
Calves that are weaned and placed on grass or small grain pastures for a period of time before finishing in a feedlot are referred to as stocker calves. There are several implants available for stocker calves (Table 3). Implant research trials have shown an improvement in the ADG of stocker cattle from 8% to 20%. Numerous trials indicate that producers can expect a 10% to 15% (0.18 pounds per day to 0.27 pounds per day) improvement in ADG over nonimplanted calves. These studies were conducted through various lengths of time, but the average was approximately 150 days, which is a typical stocker grazing period. Research results are inconclusive concerning the value of one type of product over another.
Payout for stocker implants is generally in the range of 80 days to 120 days, although several products are available with much longer payout periods. Re-implanting stockers should be considered when grazing periods are longer than the payout period (Table 3) and expected ADG during the second phase of the grazing period is moderate or high. Re-implanting in these situations has produced 4% to 6% improvement in ADG over a single implant. Alternatively, implants designed for a longer payout time may be used as the initial and only implant to provide an active implant through a long grazing season. This would eliminate the need to gather and process the animals at the midpoint of the grazing season.
Factors affecting stocker response to implants are numerous and include sex, weight, genetic gain potential, forage availability, diet quality, supplementation and environmental conditions. Research has shown that as ADG of nonimplanted controls increases (due to pasture quality or other factors), the response to an implant also increases. No adverse effects have been documented from implanting cattle that gain at very low rates. Figure 1 shows the implant response of steers stocked on wheat pasture at either 0.75 acres per steer (1.5 steers per acre; Low Gain) or 1.5 acres per steer (High Gain) (Williamson et al., 2014). Steers stocked for a low rate of gain without implants gained only 1.4 pounds per day, while implanting increased gains by 0.28 pounds per day, a 20% increase in gain with implants. Steers stocked on wheat pasture for a high rate of gain had average daily gains of 2.19 pounds per day without implants, which was increased by 0.34 pounds per day to 2.53 pounds per day with the implant, a 15% increase in gain. So, this research shows the increase in gains were similar even though the potential gains on pasture were quite different.
Research has indicated that responses to implants, supplementation and ionophores are 100% additive in stocker cattle, and there may be a slight synergistic relationship. Full benefit should be expected from both the implant and the supplement program if both are used in stocker cattle. Figure 2 shows the gain response of steers grazing wheat pasture in the fall and winter to both growth-promoting implants and an ionophore (Beck et al., 2014). Steers in this experiment that were not supplied the ionophore nor given an implant gained 2.29 pounds per day, implanted steers not fed the ionophore gained 2.71 pounds per day; an increase of 0.42 pounds per day. Feeding an ionophore increased gains by 0.20 pounds per day whether they were implanted or not, so steers fed an ionophore and implanted gained 0.6 pounds per day more than steers not implanted or fed an ionophore, which increased net returns by $42 per steer.
Figure 1. Implant response by steers grazing wheat pasture stocked to achieve either a low (1.5 steers per acre) or high (1.5 acres per steer) rate of gain during the winter and early spring. (adapted from Williamson et al., 2014)
Figure 2. Gain response of steers grazing wheat pasture to growth promoting implants and an ionophore (monensin). Adapted from Beck et al., 2014.
Reimplanting
Steers grazing native range at the Klemme Range Research Station near Bessie, Oklahoma were implanted with a combination implant supplying trenbolone acetate/estradiol (TBA/E) at receiving 60 days before grazing turnout on May 22. Steers were either not re-implanted or were re-implanted with a combination implant supplying TBA/E or estradiol and progesterone on July 23 (day 62 of grazing) at the end of the expected payout period of the previous implant (Grigsby, unpublished data). Table 5 shows the performance of the steers during the late summer grazing period from July 23 to September 30. Re-implanting with estradiol and progesterone following an initial combination TBA/E implant provided no additional gain compared with controls that were not re-implanted. Yet reimplanting with a combination implant supplying TBA/E combination increased average daily gain by 0.5 pounds per day compared with the non-re-implanted controls and by 0.9 pounds per day compared with the steers re-implanted with estradiol/progesterone. This shows the value of re-implanting following the payout period of the previous implant (in contrast with re-implanting prior to end of payout period shown in Table 6) as well as the importance of following the standard recommendation to follow initial implants with implants of equal or higher potency to see continued performance responses.
All implants are designed to release the compounds slowly through time into the bloodstream
of the animal. Different implants are formulated to provide different lengths of time
for all of the compounds to be released. This effective period or lifespan of the
implant is commonly referred to as the “payout” period. Label claims of payout range
from 60 days to 400 days. Factors that affect payout include formulation of the implant,
proper administration of the implant and blood flow to the ear. Re-implanting provides
longer-term benefits, but re-implanting before the end of the payout period of the
previous implant has not provided any additional effectiveness. Research conducted
at the USDA ARS Southern Plains Experimental Range near Fort Supply, Oklahoma shows
this relationship (Table 6). Steers given an implant supplying estradiol and progesterone
before turnout onto native range in the early summer did not respond to additional
implants given in mid-summer (day 61 of grazing). Control steers not receiving an
additional implant gaining 1.7 pounds per day and steers receiving either Estradiol
and Progesterone or a combination implant supplying Trenbolone Acetate/Estradiol gaining
1.8 pounds per day during the late summer (Grigsby, unpublished data). This research
shows re-implanting before the end of the payout period of the previous implant provides
little to no benefit.
Table 5. Effect of re-implanting grazing steers previously implanted with a trenbolone acetate/ estradiol combination (TBA/E) implant in mid-summer after implant payout on late summer performance.
| Re-implant type | ||||
|---|---|---|---|---|
| Controla | Est/Progb | TBA/Ec | P-value | |
| Steer weight, lbs | ||||
| July 23 | 667 | 667 | 666 | 0.99 |
| September 30 | 806 | 788 | 834 | < 0.01 |
| Weight gain, lbs/steer | 165.6 | 138.0 | 200.1 | 0.01 |
| Average Daily Gain, lbs/day |
2.4 | 2.0 | 2.9 | 0.01 |
Adapted from Grigsby et al. unpublished data.
a All steers were implanted with 40 mg trenbolone acetate and 8 mg estradiol at initial
processing during receiving prior to grazing. Controls were not reimplanted.
b Est/Prog – supplied 200 mg progesterone and 20 mg estradiol benzoate at reimplant.
c TBA/E – supplied 40 mg trenbolone acetate and 8 mg estradiol at reimplant.
Table 6. Effect of reimplanting grazing steers previously implanted with an estradiol/progesterone implant in mid-summer before implant payout on late summer performance.
| Re-implant Type | ||||
|---|---|---|---|---|
| Control | Est/Proga | TBA/Eb | P-value | |
| Steer weight, lbs | ||||
| July 18 | 596 | 599 | 598 | 0.80 |
| September 27 | 708 | 719 | 721 | 0.37 |
| Weight gain, lbs/steer | 113.3 | 119.7 | 121.6 | 0.12 |
| Average Daily Gain, lbs/day |
1.7 | 1.8 | 1.8 | 0.12 |
Adapted from Grigsby et al. unpublished data.
a Est/Prog – supplied 200 mg progesterone and 20 mg estradiol benzoate.
b TBA/E – supplied 40 mg trenbolone acetate and 8 mg estradiol.
Finishing Cattle
Implants are used extensively by the feeding industry in the U.S. to improve average daily gain (ADG) and feed efficiency. The finishing period can range from 120 days to 240 days. A single implant may improve ADG by 0.35 pound per day in steers and 0.25 pound per day in heifers. Feed conversion may be improved by 0.5 pounds of feed per pound of gain. Aggressive feedlot implant programs can result in up to a 21% improvement in daily gain and an improvement in feed conversion up to 11%. Maxwell et al. (2015) reported growth-promoting technologies (implanting, ionophores and feed grade antibiotics) during finishing increased body weight at harvest from 1,188 for all natural to 1,305 pounds (a 117-pound increase) due to increased ADG of 0.88 pounds per day (from 2.62 pounds per day for all-natural to 3.48 pounds per day for conventional), improving feed efficiency by 21% (8.33 pounds vs 6.57 pounds of feed per pound of gain). Hot carcass weights were increased by 84 pounds with growth promoting technologies (from 767 pounds hot carcass weight [HCW] with all-natural to 850 pounds HCW with conventionally produced calves). This increased efficiency and weight gain produces a significant economic return, reduces the resources needed to produce beef and decreases the environmental footprint of beef production.
An implant program for finishing cattle must evaluate numerous factors, including
decisions concerning timing of implant, type and amount of hormone activity and number
of implant times. There are many different options available for implanting finishing
calves (Table 4) ranging from very conservative (low-potency hormone levels) to very
aggressive (high-potency hormone levels). Selection of implant program depends on
previous management history, genetics of the animal and production and marketing goals.
Implants can have pronounced effects upon carcass characteristics of cattle. In general,
when cattle are fed the same number of days, implants improve carcass weight and ribeye
area, while decreasing marbling scores. With these circumstances, implants may reduce
the percentage of cattle grading at least USDA Choice by 2% to 24% (Duckett and Owens,
1997). Implants may slightly increase skeletal maturity, which also impacts USDA Quality
Grade. Type of implant, gender and genotype of the animal all influence these responses.
However, if cattle are harvested at constant back fat thickness, implants may have
little to no impact on quality grade.
For a complete review of implant effects during finishing, see Duckett and Owens, 1997.
Lifetime Implanting Strategies
In the modern beef industry, it is fairly common for cattle to receive three or more implants during their lifetime. For producers who operate in only one segment of the industry, the implant decision is simple. However, for producers who retain ownership of an animal through two or more phases and market cattle on a carcass merit price grid, implant decisions become more complex. It is possible that implants administered in one phase can have carryover effects in subsequent phases, however in many studies, this carryover effect has not materialized (Reuter and Beck, 2013). Implants approved for suckling calves are less potent than those approved for stockers, which are less potent than many feedlot implants. A strategy to maximize lifetime gain of the animal while minimizing deleterious effects on carcass quality and animal behavior is an implant program using increasingly potent implants. During the suckling phase, a low-potency implant will be used, followed by one or two moderate implants in the growing phase, followed by a moderate implant upon placement in the feed yard, then a high-potency implant 80 days to 100 days before slaughter. The effects of multiple implants on marbling scores may become more dramatic as three or more implants are used during the animal’s lifetime.
Barham et al. (2012) finished two sets of calves either directly after preconditioning for 63-days (calf-fed) or as yearlings following an extensive 133-day low growth stocker period. Calves were managed with either aggressive implanting (implants administered at weaning, during grazing [yearlings only], at arrival to the feedlot and re-implanted during finishing) or delayed implanting (implants administered only during finishing). Breeding selection for the herd of one set of calves had been for carcass quality and growth, while the herd for the second set were selected for maternal hybrid vigor and growth traits. The aggressive implant program increased growth and hot carcass weights of both calf-feds and yearlings from both herds. In the first herd (the one selected for carcass quality) aggressive implanting program reduced marbling score of both calf-feds and yearlings, and the impact was greatest in yearlings with the percentage of USDA Choice quality grade or greater was decreased from 95% in delayed implant to 45% in aggressively implanted cattle. While in the second herd (not selected for carcass quality) marbling score, USDA quality grade and the percentage USDA Choice was not affected by an implant program. This research indicates that aggressively implanting cattle prefinishing with high genetic propensity for marbling during a period of restricted nutrition can have a large impact on subsequent carcass quality, yet marbling and carcass quality of cattle with limited genetic selection for those traits are not affected by implant program prefinishing.
Producers who retain ownership of animals through more than one production phase should
evaluate their overall implant program for the way they are marketing their cattle.
Factors to consider are the feed cost, the base value of additional carcass weight,
the Choice-Select spread and the potential value of marketing cattle into specialty,
non-hormone treated cattle (NHTC) programs.
Economics
Implants are one of the most cost-effective technologies available to cattle producers. Stocker implants typically return more than $15 for every $1 invested. Implants effectively increase growth rate, increase protein deposition and improve feed efficiency resulting in approximately a 7% overall reduction in the cost to produce beef (Lawrence and Ibarburu, 2006). Consider this example of the economic potential of implanting calves who are still nursing their mothers: A nursing calf, implanted at 3 months of age and 150 days before weaning may gain an additional 0.10 pound per day for 150 days. The 15 pounds additional weaning weight could have a value of $1.00 per pound to $2.00 per pound for a total of $30. The implanting cost is approximately $0.85. Here, the net return would be $29.15 per calf sold.
Alternative production systems such as organic, NHTC or “natural,” generally do not
allow cattle to be implanted. Producers who want to use these production systems should
ensure the premiums they receive for these cattle will offset the reduced production
and efficiency that implants (and other technologies) offer. Historically, these premiums
have not been adequate to offset the lost production (Maxwell et al., 2015). Based
on research comparing all-natural NHTC-raised calves using no growth promoting technologies
and conventionally produced calves managed using implants and ionophores (Maxwell
et al., 2015), the all-natural calves were 115 pounds lighter at harvest (1,188 pounds
vs 1,305 pounds), had lower ADG (2.6 pounds per day vs 3.5 pounds per day) and had
84 pound lighter hot carcass weights (767 pounds vs 851 pounds) with no difference
in percent USDA Choice quality grade (90% vs 91%). All natural premiums for the NHTC
calves would have to be $11/cwt at slaughter based on a $114/cwt live cash market
in order for breakeven from the lost production of forgoing the use of growth-promoting
technologies. Beck et al. (2012) found that with typical USDA Choice-Select price
spread ($8/cwt of HCW) implanting throughout the preconditioning, stocker, and finishing
phases increased net returns by $35 per head to 70 per head compared with implanting
during the late finishing period only.
Beef Quality Assurance
Implant Location
The only approved implantation site for all brands of implants is subcutaneously in the middle one-third of the back of the ear. The implant must not be closer to the head than the edge of the auricular cartilage ring farthest from the head. The procedure to insert the implant should be done under conditions as sanitary as possible. Cleaning the ear, keeping equipment clean and using a sharp needle are all recommended. Problems with ear abscesses are the most common cause of implant defects and are usually related to poor sanitation while implanting. Proper animal restraint makes the implanting placement more accurate and the procedure safer for the handlers. Follow all manufacturers’ recommendations for implant administration.
Figure 3 shows the correct location. The Food and Drug Administration (FDA) no longer allows implants to be placed at the base of the ear.
Figure 3. The correct location for implants.
Implanting Procedure
A qualified and trained individual should be assigned the task of implanting. Employing the following steps will greatly diminish the incidence of implanting errors, such as abscesses, crushed pellets or missing implants. Achieving an active, undamaged uncontaminated implant in each calf is the goal. Speed will come with practice; it is better to do it right the first time than to have to go back and fix mistakes. Similarly, it is much better to take time and do it right than to pay for an implant and not realize a $20 per head to $30 per head advantage because the job was not done correctly.
- Read the label for all animal health products. Ensure the correct dosage, location and procedures are followed. Ensure the product is labeled for use in the class of animal to which it is being administered. Any deviation from label directions carries the potential for stiff legal penalties.
- Obtain all of the necessary equipment to maintain sanitation. A tray and large sponge
soaked in a disinfectant should be used to store the implant applicator between animals.
An extra needle for the applicator should be available in case the needle becomes
dull, burred, bent or broken. A clean table out of the way of flying debris should
be used to store the applicator and implants between uses. (Figure 4)
Figure 4. Storing the applicator and implants between uses.
-
Become familiar with the operation of the implant applicator.
- Properly restrain the animal to prevent movement (Figure 5). If necessary, further
restrain the head by use of a halter.
Figure 5. Properly restrain the animal.
- Inspect the animal’s ear. Check for previous implants or abscesses, presence of ear
tags or ear tag holes, mud, manure or other debris. Clean and dry the implant area
by scraping with a knife blade or by wiping with a paper towel and disinfectant (Figure
6). Do not attempt to implant through mud or manure. If an implant is present do not
re-implant.
Figure 6. Clean the area with a knife blade or disinfectant
-
If necessary, wipe off hands before handling the applicator. Mud, manure and blood can contaminate the inner workings of the applicator.
-
Wipe the needle through the sponge to disinfect it. Pull the tip of the needle across the sponge with the bevel facing down against the sponge to clean out any material inside the needle (Figure 7).
Figure 7. Clean the needle.
- Pinch the tip of the animal’s ear between the thumb and index finger of the left hand
(for the left ear). Place the tip of the applicator needle against the ear at a slight
angle, bevel side up or away from the ear, at the outer edge of the implant zone (Figure
8).
Figure 8. Place the needle at an angle at the outer edge of the implant zone.
-
Slide the needle under the skin of the ear and insert it fully. Make sure it is under the skin and not in the cartilage or punctured all the way through the ear. If the needle skips off the back of the ear, return to step number 7. Mud or other debris likely will have gotten caught in the needle bevel, and if not cleaned will be implanted into the ear with the implant on the next attempt. Using sharp needles and slowing down can reduce skipping off.
-
Slide the needle back out of the ear about as far as the length of the implant. Some models of implant applicators have needles that automatically withdrawal the needle.
-
Pull the trigger to deposit the implant and withdraw the needle completely.
-
Feel the implant site to ensure the pellets were correctly deposited, not bunched up or crushed (Figure 9). If so, check equipment, properly restrain the animal and slow down.
Figure 9. Feel the implant to ensure it is not bunched up or crushed.
- Return the applicator to the tray and wipe across the sponge to disinfect it (Figure 7).
Other Best Practices
- Consult and follow label for all products used.
- Implants have no slaughter withdrawal, as the ear is always removed as offal during the slaughter process.
- No implants are cleared for use in classes of cattle besides calves, stockers and feedlot animals. This includes breeding animals, cull cows, dairy cattle and veal calves.
- Implants should not be administered at birth due to hormonal development of the calf. Some label instructions specify a minimum of 30 days (Ralgro®) or 45 days (Synovex®-C and Component® EC with Tylan®) of age for administration of calf implants, depending on the implant.
- Implants should be stored properly to maintain effectiveness. Store in a clean dry place in a plastic bag sealed to keep out moisture and debris. Consult the label for storage conditions and time of storage after opening.
- If possible, implant cattle on dry days when the cattle are dry and free of mud. This will reduce the incidence of abscesses.
- One implant manufacturer offers a line of implant products that include both the anabolic compound pellet and a pellet containing a dose of the antibiotic Tylan®. The purpose of the antibiotic pellet is to dissolve soon after administration and reduce the incidence of implant site abscesses.
Animal Health
Implants are suspected to directly cause, or be associated with, several undesirable changes in animals. Responses normally associated with reproductive processes are observed in heifers, including signs of estrus, vaginal or rectal prolapses, development of the udder and other problems. Implants may increase the incidence of bullers in steers. Bullers are steers that mount others or will stand to be mounted similar to the behavior of a cow in estrus. However, it is thought that bulling is caused by a physiological defect in the animal and implants merely exacerbate this condition. Estimates of the frequency of the occurrence of bullers range from 1% to 4%.
References
Barham, B., P. Beck, S. Gadberry, J. Apple, W. Whitworth, and M. Miller. (2012) Effect of age entering the feedlot and implant potency on animal performance, carcass quality, and consumer acceptance of beef. The Professional Animal Scientist Vol. 28. Pg. 30.
Beck, P. B. Barham, J. Apple, W. Whitworth, M. Miller, and S. Gadberry. (2012) Effect of age entering feedlot and implant regimen on finishing system profitability. Professional Animal Scientist. Vol. 28. Pg. 32.
Beck, P. A., T. Hess, D. Hubbell, D. Hufstedler, B. Fieser, and J. Caldwell. (2014) Additive effects of growth promoting technologies on performance of grazing steers and economics of the wheat pasture enterprise. Journal of Animal Science. Vol. 92. Pg. 1213.
Duckett, S. and F. Owens. (1997) Effects of implants on performance and carcass traits of feedlot steers and heifers. Proceedings: Impact of Implants on Performance and Carcass Value of Beef Cattle. Oklahoma State University, P-957. Pg. 63.
Johnson, R., D. Doye, D. Lalman, D. Peel, and K. Raper. (2008) Adoption of best management practices in stocker cattle production. Selected Paper prepared for presentation at the Southern Agricultural Economics Association Annual Meeting, Dallas, TX, February 2-6, 2008.
Lawrence, John D and Maro A. Ibarburu. (2006) Economic analysis of pharmaceutical technologies in modern beef production. Iowa State University. http://econ2.econ.iastate.edu/faculty/lawrence/documents/GET7401-LawrencePaper.pdf
Maxwell, C. L., B. C. Bernhard, C. F. O’Neill, B. K. Wilson, C. G. Hixon, C. L. Haviland, A. N. Grimes, M. S. Calvo-Lorenzo, D. L. VanOverbeke, G. G. Mafi, C. J. Richards, D. L. Step, B. P. Holland, C. R. Krehbiel. (2015) The effects of technology use in feedlot production systems on feedlot performance and carcass characteristics, Journal of Animal Science, 93(3):1340–1349, https://doi.org/10.2527/jas.2014-8127.
Preston, R. (1997) Rationale for the Safety of Implants. Proceedings: Impact of Implants on Performance and Carcass Value of Beef Cattle. Oklahoma State University, P-957. Pg 199.
Reuter, R. R. and P. A. Beck. (2013) Southern Section Interdisciplinary Beef Symposium.: Carryover effects of stocker cattle systems on feedlot performance and carcass characteristics. Journal of Animal Science. Vol 91, Pg. 508.
USDA-NAHMS. (2013) Feedlot 2011 Part IV. Health and health management on U. S. Feedlots with a capacity of 1,000 or more head. National Animal Health Monitoring System, United States Department of Agriculture. https://www.aphis.usda.gov/animal_health/nahms/feedlot/downloads/feedlot2011/Feed11_dr_PartIV_1.pdf
Williamson, J. A., R. R. Reuter, J. A. Apple, C. B. Stewart, H. C. Gray, and P. A. Beck. (2014) Growth promoting implants and nutrient restriction prior to feeding: effect on finishing performance, carcass composition, carcass quality, and consumer acceptability of beef. The Professional Animal Scientist. Vol. 30. Pg. 485.
Vestal, M., C. Ward, D. Doye, and D. Lalman. (2007) Cow-calf Production Practices in Oklahoma – Part I. OSU Extension Fact Sheet AGEC-245, Oklahoma Cooperative Extension Service, Oklahoma State University.

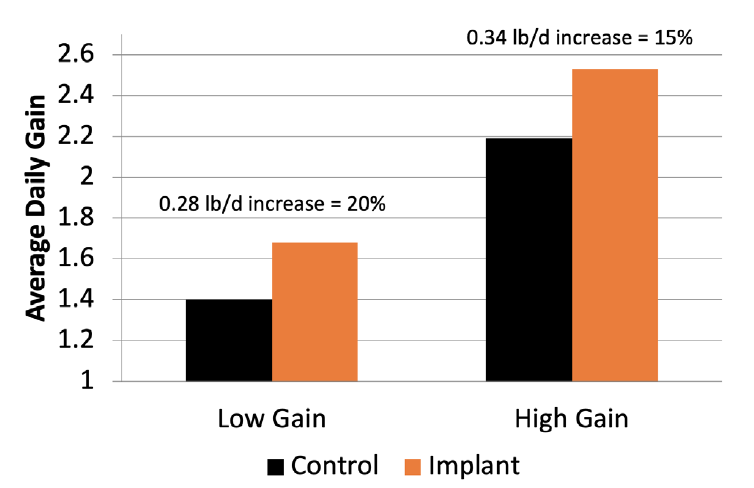
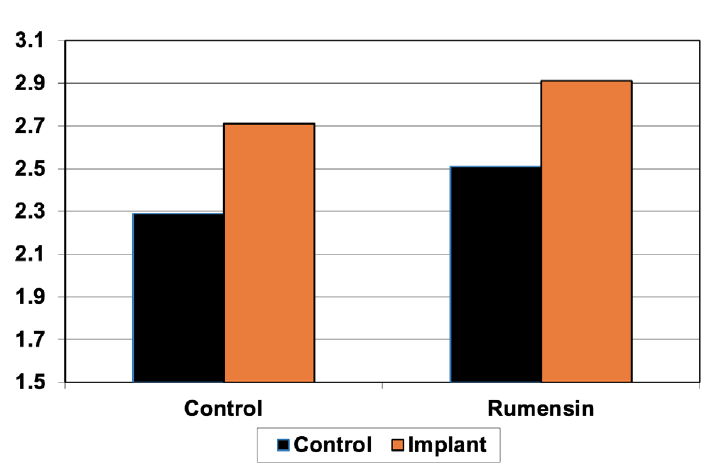
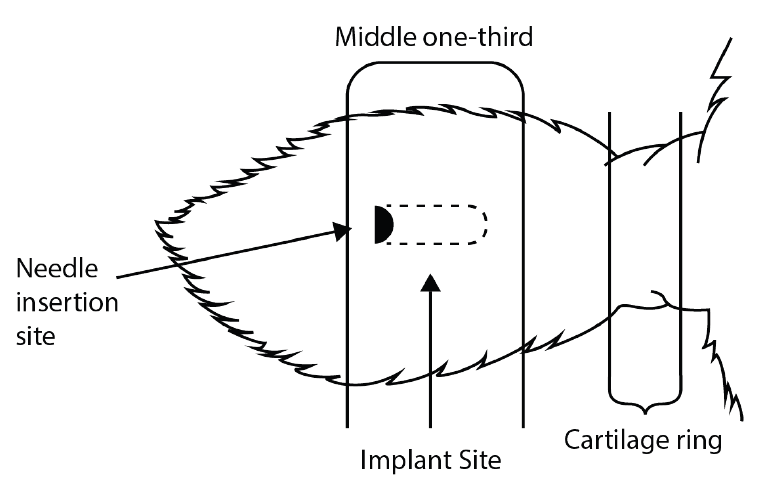
 Figure 4. Storing the applicator and implants between uses.
Figure 4. Storing the applicator and implants between uses. Figure 5. Properly restrain the animal.
Figure 5. Properly restrain the animal.
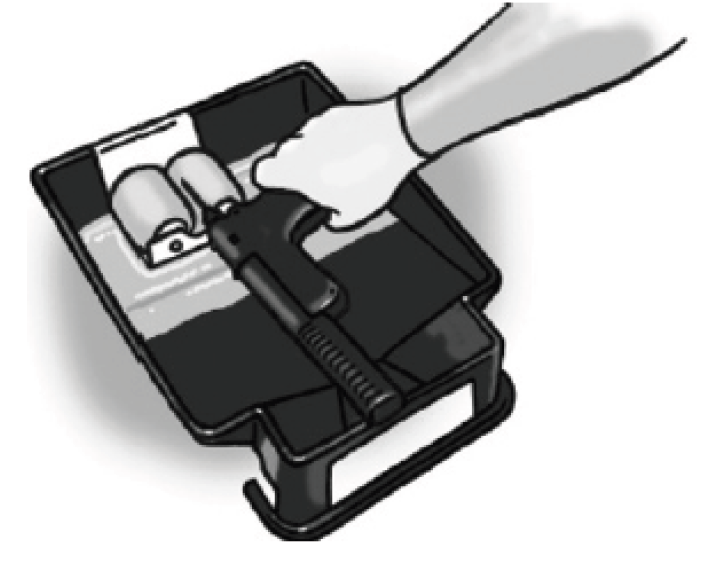 Figure 7. Clean the needle.
Figure 7. Clean the needle.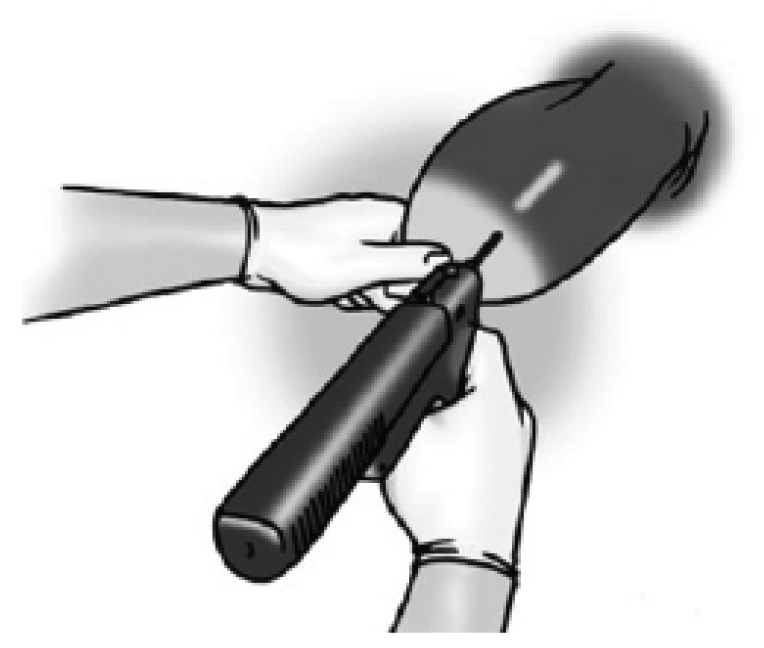 Figure 8. Place the needle at an angle at the outer edge of the implant zone.
Figure 8. Place the needle at an angle at the outer edge of the implant zone.  Figure 9. Feel the implant to ensure it is not bunched up or crushed.
Figure 9. Feel the implant to ensure it is not bunched up or crushed.