Soilborne Diseases of Peanut
The agents (pathogens) that cause soilborne diseases of peanut are various fungi and nematodes that survive in the soil and infect one or more parts of the plant at or below the soil surface. Soilborne diseases can severely limit peanut yields in Oklahoma, particularly in fields where long rotations with non-host crops are not practiced. Southern blight and Sclerotinia blight are probably the most widespread and destructive of the soilborne diseases and are described in Extension Fact Sheet EPP-7186. There are several other soilborne diseases that are less common, but still important where they occur. The purpose of this fact sheet is to describe these diseases to aid growers in their identification and to provide general suggestions for their management. Specific suggestions for cultural and chemical controls are updated yearly in the OSU Extension Agents Handbook of Insect, Plant Disease, and Weed Control (Circular E-832) and in the Oklahoma Peanut Production Guide (Circular E-608).
Aspergillus Crown Rot
Aspergillus crown rot commonly results in seedling blight, but also may affect older plants from mid- to late-season. The fungus that causes crown rot, Aspergillus niger, is different from the species that causes aflatoxin contamination of harvested kernels. A niger is present in most peanut soils and is a common contaminant of peanut seed. However, outbreaks of the disease are sporadic and appear to be related to the prior occurrence of one or more stresses. Extreme heat or fluctuations in soil moisture during the seedling stage, poor seed quality, seedling damage from pesticides or cultivation, and feeding by root and stem boring insects are stresses thought to aggravate the disease. The economic importance of Aspergillus crown rot is difficult to assess. Generally scattered plants are affected, although stand losses of 50 percent have been reported in isolated fields.
Symptoms
While the fungus may cause seed rot and pre-emergence damping-off of seedlings, the most obvious symptom is the sudden wilting of young plants. The crown area of infected plants just below the soil line may be swollen and eventually becomes covered with a black, sooty mass of fungus growth (Figure 1). Most affected plants die within 30 days of planting. Later in the season, individual branches or entire plants may develop similar symptoms. Splitting the crown and tap root of affected plants reveals an internal discoloration of the vascular system that is dark grey in color.

Figure 1. Aspergillus crown rot with characteristic black fungus (mycelium) growing on the root surface.
Control
There are no effective controls known for Aspergillus crown rot. All commercially grown varieties are susceptible. However, precautions can be taken that may reduce disease severity. Plant high quality seed treated with a fungicide. Maintain adequate and uniform soil moisture and avoid throwing soil against plants during cultivation. Root and stem boring insects such as the lesser cornstalk borer, which may aggravate the disease, should be controlled where warranted (See Extension Fact Sheet EPP-7174).
Black Hull
Black hull is a common pod disease in Oklahoma and is most severe on Spanish varieties. In New Mexico, black hull has long been a serious problem on valencia peanuts grown for the in-shell market. The black hull fungus, Theilaviopsis basicola, also causes black root rot of cotton seedlings, alfalfa, and several other broadleaf crops.
The fungus is long-lived in soil and survives by producing resistant spores. Factors that promote black hull development are high soil pH (7.0 and above), low temperatures late in the growing season, excessive soil moisture, and crop rotations with susceptible crops.
Symptoms
Pod symptoms are scattered black dots or large blackened areas on the pod surface (Figure 2). Usually the discoloration is superficial, but the decay may extend into the pod causing kernel discoloration. Under severe disease pressure, numerous black spots (lesions) form on the pegs resulting in pod loss during harvest.
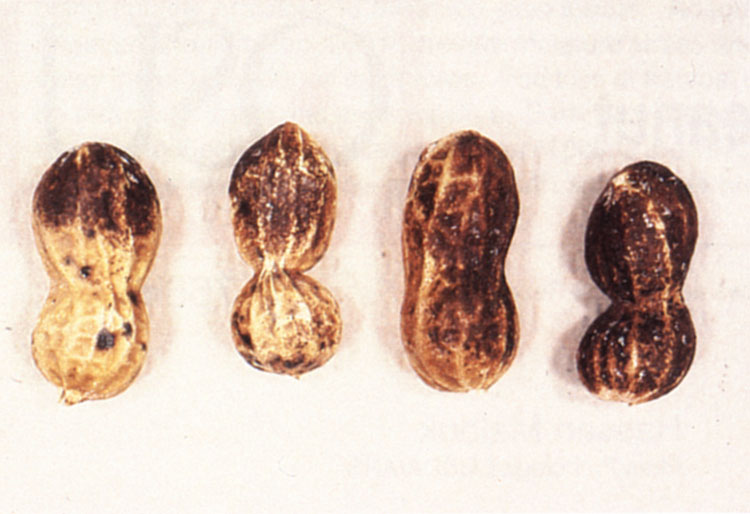
Figure 2. Superficial black discoloration of pods caused by black hull.
Control
No commercial peanut varieties are resistant to black hull, although some Spanish and valencia varieties appear highly susceptible and should be avoided in problem fields if possible. Avoid excessive irrigation and crop rotations with cotton and alfalfa. Crop rotation with grain sorghum is used to reduce black hull in New Mexico.
Limb Rot
Limb rot is the foliar blight phase of a disease complex in peanut caused by the fungus Rhizoctonia solani. This fungus also causes seedling disease and root, pod, and peg rots. Limb rot is usually of minor importance in Oklahoma, but causes problems in the production of runner varieties during wet periods late in the season. Limb rot is favored by frequent rains, excessive vine growth, and mechanical damage to vines.
Symptoms
Limb rot usually first appears in late August and September after the vines of runner varieties have lapped. Elongated spots (lesions) develop on the underside of lower branches in contact with the soil. The lesions are dark brown and may have a target-like appearance (Figure 3). Lesions expand and girdle individual branches causing them to wilt and die. Several branches may be killed, but generally not entire plants as with Sclerotinia and southern blights. In severe cases where rainfall is excessive, leaves within the canopy are covered with a white, moldy growth. In addition to causing pod rot (see Pod Rot below), the fungus also may infect the tips of pegs as they penetrate the soil. The pod-forming region of infected pegs is killed, turns black, and appears shredded.

Figure 3. Limb rot lesion on a peanut stem with dark concentric rings. (Courtesy T.A. Lee, Texas A & M University)
Control
Cultural and chemical controls are effective against this disease. Spanish varieties are resistant to limb rot because of their upright growth habit. For runner varieties, avoid excessive fertilization and irrigation which promote rank vine growth and create an ideal environment for disease development. Minimize mechanical injury to the vines where possible. Preventive treatment with one of the fungicides effective against southern blight also provides limb rot control. These fungicides also may help control the peg rot phase of the disease.
Nematode Diseases
Nematodes are microscopic, round worms that live in soil. The species of nematodes that cause the most damage to peanuts in Oklahoma are the northern root-knot, peanut root-knot and root-lesion nematodes. The northern root-knot nematode is the most widespread. The peanut root-knot nematode is less common, but very damaging where it occurs. The root-lesion nematode is common, but is less damaging than the root-knot nematodes (northern and peanut).
Severe nematode infestations may develop with intensive peanut production. High populations of root-knot and root lesion nematodes can reduce plant growth, pod production, and directly damage pods. Nematodes thrive in sandy soils where damage is usually greatest. Nematode feeding on pods also may also increase levels of pod and kernel rots.
Symptoms
Plants infected with the northern root-knot nematode have a dense, bushy root system (Figure 4) with tiny galls (swellings) apparent at the junctions of root branches. Larger galls also may form on pegs and pods under heavy infestations. Galls from the peanut root-knot nematode are larger and easier to recognize, causing the roots to swell up to ½ inch in diameter and appear corky in texture (Figure 5). Where above-ground symptoms of root-knot infection are apparent, varying degrees of plant yellowing and stunting occur. Above-ground symptoms are generally most severe in sandy soils where populations are high. Stunted, off-color plants usually occur in clumps along the row, resulting in uneven row growth (Figure 6). Pod set can be drastically reduced under severe root-knot pressure. Where peanut root-knot nematode is severe, leaves, branched, and/or entire plants may be killed.
Symptoms of root-lesion nematode occur mainly below ground, although poor plant growth may also be caused by this nematode. Dark brown spots develop on roots and pegs of infected plants. Numerous brown, pinpoint-size spots appear on infected pods. Spots eventually darken and enlarge, leaving a peppered appearance to the pods (Figure 7). Nematode feeding also may weaken the pegs resulting in pod loss at harvest.

Figure 4. Left – Stunted plants with bushy root systems caused by the northern root-knot nematode, Right – healthy plants.

Figure 5. Large galls and swollen root system caused by the peanut root-knot nematode.

Figure 6. Clumped distribution of stunted plants caused by the northern root-knot nematode.

Figure 7. Pod speckling caused by the root-lesion nematode.
Control
Management strategies differ depending on the specific nematode present and the level of infestation. Therefore, identification of the specific nematode present within a field and determination of its population level are essential for developing an effective control program. Because it is impossible to eradicate nematodes from fields, the goal of a control program is to reduce populations below damaging levels. Known or suspected problem fields should be sampled each year before planting a peanut crop. Samples should be submitted to a diagnostic laboratory for accurate identification and population assessment. Consult your county Extension educator and/or Extension Fact Sheet EPP-7610 for information on nematode sampling and submitting samples to the OSU Plant Disease Diagnostic Laboratory.
Root-knot nematodes can be effectively managed with crop rotation. Cotton, corn, grain sorghum, and sudan grass are excellent rotation crops for reducing populations of this nematode. Avoid rotations with root-knot hosts such as soybean, alfalfa, vegetable crops, and forage legumes. Where potentially damaging nematode populations exist and peanuts are to be planted, the use of a nematicide should be considered. Peanut varieties with good resistance to the peanut root-knot nematode, but not the northern root knot nematode are available.
Root-lesion nematodes are more difficult to control than root-knot nematodes. Crop rotation has not been effective in reducing levels of this species because it has a wide host range and can reproduce on most rotational crops including grasses. Treatment of a problem field with a nematicide might be beneficial.
Pod Rot
Pod rot is a term used to describe the decay of pods in soil. There are several causes of pod decay and discoloration in peanuts. Southern blight (see Extension Fact Sheet EPP-7186) is a leading cause of pod rot in Oklahoma. Nematodes and black hull also cause pod discoloration, but the decay is usually superficial. The specific pod rots described here are caused by the fungi Pythium spp. and Rhizoctonia solani which are often found together in rotted pods. Advanced stages of Pythium and Rhizoctonia pod rots result in complete decay of the pod and kernels. Yield losses can be substantial where a large percentage of the pods are affected.
The fungi that cause pod rot are normally found at some level in most peanut soils. Likewise, plants with a few rotted pods are found in most fields. Severe outbreaks of pod rot are less common, but can be devastating where they occur. Pod rot tends to be severe in sandy soils and in fields intensively cropped to peanuts. A complex of factors in addition to the two fungi are probably responsible for these severe outbreaks. These factors include excessive soil moisture, wide fluctuations in soil moisture, calcium deficiency, insect and nematode feeding, and irrigation with poor quality (salty) water.
Symptoms
No apparent symptoms of pod rot appear above ground except that plants with severe pod rot may flower profusely and appear abnormally dark green in color late in the season. Below ground, symptoms of pod rot caused by Rhizoctonia and Pythium are difficult to distinguish because both pathogens are often present. Light brown areas develop on pods which later turn dark brown or black (Figure 8). A few to nearly all of the pods on a plant may be affected. Pythium usually causes a black, watery rot. Pods rotted by Rhizoctonia have a firm brown decay, and the seeds and inner pod wall may be lined with a cream-colored fungus. The seeds within rotted pods are usually completely decayed or severely damaged.

Figure 8. Various stages of Pythium and Rhizoctonia pod rot.
Control
Because pod rot is difficult to control, management strategies are aimed at reducing losses. Fields with a history of pod rot should be identified. A crop rotation program with crops such as grain sorghum, corn, and sudan grass should be implemented. Avoid planting susceptible varieties such as Virginia types and non-adapted runner varieties in problem fields. Spanish varieties are most resistant to pod rot. Nematodes and pod-feeding insects should be controlled where they are a problem. Soils deficient in calcium should be corrected. Application of gypsum as a band over the row at flowering has been beneficial in some production areas. However, most soils in Oklahoma are high in calcium, and gypsum application generally has not been effective. Applications of fungicides containing metalaxyl (Ridomil), azoxystrobin (Abound), and phosphorous acid at pegging have provided partial control of pod rot.
Phymatotrichum Root Rot
Phymatotrichum root rot is also known as cotton root rot or Texas root rot. In addition to peanut, the fungus (Phymatotrichum omnivorum) attacks a wide range of broadleaf crops including cotton and alfalfa and survives in the soil indefinitely. In Texas, the disease is widely distributed in blackland soils where cotton is a primary cash crop and the disease has resulted in reduced land values. In Oklahoma, the disease is limited in distribution along the Red River. The fungus thrives in alkaline soils, but has been found recently in a few fields with a neutral soil pH. The disease is very destructive, resulting in total yield loss within infested areas of the field.
Symptoms
Plants in scattered patches wilt and die at mid-season (Figure 9). The taproot of wilted plants is decayed, and the internal vascular system near the crown is brown in color. Infected areas increase in size as newly wilted plants appear at the leading edge of the patches. Patches may eventually reach up to 1/2 acre in diameter. Following cloudy and rainy weather, tan- to buff-colored patches of fungal growth (spore mats), 6 to 12 inches in diameter, may appear on the soil surface near the leading edge of the infection centers. These spore mats are transient and dissipate after a few days. The spores from these mats are not capable of infecting plants. New infections arise from the fungal strands that grow through the soil. New roots may form just below the soil surface on plants in the center of the patches resulting in some regrowth of the vines.

Figure 9. Plants killed at mid-season by Phymatotrichum root rot.
Control
There are no effective controls for Phymatotrichum root rot. Avoid planting peanuts, cotton, or alfalfa in fields infested with this disease. Long rotations with grain sorghum may reduce the severity of this disease.
Seedling disease
Seedling diseases are caused by several fungi including Pythium, Rhizoctonia, and Fusarium. These fungi are common in most agricultural soils and attack seeds and seedling of many crops. Seedling disease in peanut is favored by cool and wet soils which slow seed germination and seedling growth. Severe seedling disease may result in stand failure which necessitates replanting to avoid yield loss. Seedling disease should be distinguished from herbicide injury which also is favored by the same conditions.
Symptoms
Failure of seedlings to emerge from the soil (pre-emergence damping-off) or sudden wilt and death of seedlings shortly after emergence (post-emergence damping-off) are symptoms of seedling disease. A reddish brown to black decay of the roots, or the stem near the soil line are commonly observed on infected plants. Affected seedlings frequently wilt under hot temperatures because of the root and lower stem dysfunction.
Control
Fungicide seed treatments applied by seed dealers effectively control seedling disease in most instances. Additionally, cultural practices should be used to help insure a good stand. These include good seedbed preparation, use of a raised seedbed to promote drainage, planting high quality seed with good vigor, and planting at recommended soil temperatures and depths. In fields with unusually severe seedling disease pressure, additional fungicide may be applied as a hopper-box or in- furrow treatment at planting.
Verticillium Wilt
Verticillium wilt is caused by the fungus Verticillium dahliae which also causes wilt disease of many crop plants including cotton. The disease is a common problem in Oklahoma. The fungus persists in soil for long periods as tiny seed-like structures called microsclerotia. Fields become contaminated with Verticillium where microsclerotia have been introduced with soil or water. Microsclerotia germinate and infect nearby roots. The fungus then grows into the vascular system resulting in plant wilting. Drought stress accelerates symptom development. Significant yield losses result when plants are killed prior to maturity.
Symptoms
Wilt symptoms generally do not appear before flowering and most often develop during pod set, although plants are probably infected earlier. The upper leaves of infected plants turn pale green, curl, and then die from the leaf margins inward (Figure 10). Infected branches or whole plants become stunted, turn yellow, and may wilt and die. A tan to brown colored discoloration of the vascular system is apparent when affected leaf petioles, stems, or tap root are cut and examined (Figure 11). Well-watered plants may survive for a long period, but the disease itself accelerates maturity.

Figure 10. Foliar symptoms of Verticillium wilt.
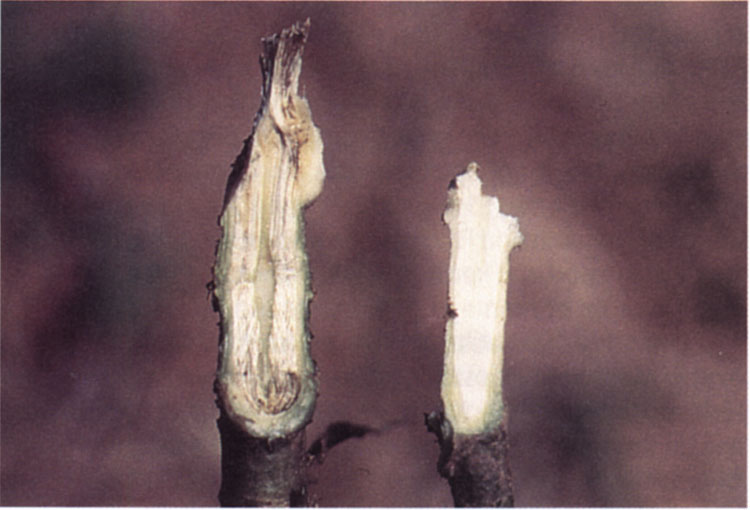
Figure 11. Right – Vascular crown discoloration caused by Verticillium wilt, Left – healthy crown.
Control
There is no effective control for Verticillium wilt. Therefore, contamination of clean fields with soil or crop debris from infested fields should be avoided. Once a field is contaminated, the disease can only be partially controlled using cultural practices to minimize yield losses. Adequate irrigation should be applied to prevent moisture stress and to delay the progression of symptoms. Pod loss at harvest can be reduced if the plants are kept alive. Early harvest may be required for fields where a large number of plants are infected early in the season. Crop rotations with susceptible crops such as cotton and potato should be avoided. Long rotations with non-hosts such as corn, grain sorghum, and sudan grass may be beneficial. Although no peanut varieties are resistant to Verticillium wilt, Spanish and valencia types are most susceptible.
John Damicone
Extension Plant Pathologist
