Recirculating Aquaculture Tank Production Systems: Aquaponics—Integrating Fish and Plant Culture
- Jump To:
- System Design
- Fish Production
- Solids
- Solids Removal
- Biofiltration
- Hydroponic Subsystems
- Sump
- Construction Materials
- Component Ratios
- Plant Growth Requirements
- Nutrient Dynamics
- Vegetable Selection
- Crop Production Systems
- Pest and Disease Control
- Approaches to System Design
- Economics
- Overview
Aquaponics, the combined culture of fish and plants in recirculating systems, has become increasingly popular. Now a news group (aquaponicsrequest@ townsqr.com — type subscribe) on the Internet discusses many aspects of aquaponics on a daily basis. Since 1997, a quarterly periodical (Aquaponics Journal) has published informative articles, conference announcements and product advertisements. At least two large suppliers of aquaculture and/or hydroponic equipment have introduced aquaponic systems to their catalogs. Hundreds of school districts are including aquaponics as a learning tool in their science curricula. At least two short courses on aquaponics have been introduced, and the number of commercial aquaponic operations, though small, is increasing.
Aquaponic systems are recirculating aquaculture systems that incorporate the production of plants without soil. Recirculating systems are designed to raise large quantities of fish in relatively small volumes of water by treating the water to remove toxic waste products and then reusing it. In the process of reusing the water many times, non-toxic nutrients and organic matter accumulate. These metabolic by-products need not be wasted if they are channeled into secondary crops that have economic value or in some way benefit the primary fish production system. Systems that grow additional crops by utilizing by-products from the production of the primary species are referred to as integrated systems. If the secondary crops are aquatic or terrestrial plants grown in conjunction with fish, this integrated system is referred to as an aquaponic system (Figure 1).
Figure 1. Nutrients from red tilapia produce a valuable crop of leaf lettuce in the UVI aquaponic system.
Plants grow rapidly with dissolved nutrients that are excreted directly by fish or generated from the microbial breakdown of fish wastes. In closed recirculating systems with very little daily water exchange (less than 2 percent), dissolved nutrients accumulate in concentrations similar to those in hydroponic nutrient solutions. Dissolved nitrogen, in particular, can occur at very high levels in recirculating systems. Fish excrete waste nitrogen, in the form of ammonia, directly into the water through their gills. Bacteria convert ammonia to nitrite and then to nitrate (see SRAC Publication No. 451, “Recirculating Aquaculture Tank Production Systems: An Overview of Critical Considerations”). Ammonia and nitrite are toxic to fish, but nitrate is relatively harmless and is the preferred form of nitrogen for growing higher plants such as fruiting vegetables.
Aquaponic systems offer several benefits. Dissolved waste nutrients are recovered by the plants, reducing discharge to the environment and extending water use (i.e., by removing dissolved nutrients through plant uptake, the water exchange rate can be reduced). Minimizing water exchange reduces the costs of operating aquaponic systems in arid climates and heated greenhouses where water or heated water is a significant expense. Having a secondary plant crop that receives most of its required nutrients at no cost improves a system’s profit potential. The daily application of fish feed provides a steady supply of nutrients to plants and thereby eliminates the need to discharge and replace depleted nutrient solutions or adjust nutrient solutions as in hydroponics. The plants remove nutrients from the culture water and eliminate the need for separate and expensive biofilters. Aquaponic systems require substantially less water quality monitoring than separate hydroponic or recirculating aquaculture systems. Savings are also realized by sharing operational and infrastructural costs such as pumps, reservoirs, heaters and alarm systems. In addition, the intensive, integrated production of fish and plants requires less land than ponds and gardens. Aquaponic systems do require a large capital investment, moderate energy inputs and skilled management. Niche markets may be required for profitability.
System Design
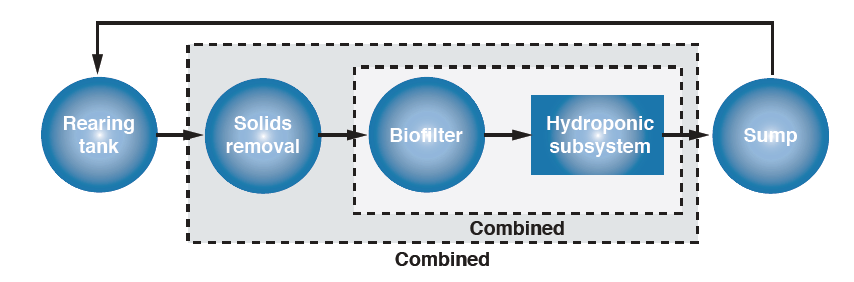
The design of aquaponic systems closely mirrors that of recirculating systems in general, with the addition of a hydroponic component and the possible elimination of a separate biofilter and devices (foam fractionators) for removing fine and dissolved solids. Fine solids and dissolved organic matter generally do not reach levels that require foam fractionation if aquaponic systems have the recommended design ratio. The essential elements of an aquaponic system are the fish-rearing tank, a settleable and suspended solids removal component, a biofilter, a hydroponic component, and a sump (Figure 2).
Figure 2. Optimum arrangement of aquaponic system components (not to scale).
Effluent from the fish-rearing tank is treated first to reduce organic matter in the form of settleable and suspended solids. Next, the culture water is treated to remove ammonia and nitrate in a biofilter. Then, water flows through the hydroponic unit where some dissolved nutrients are taken up by plants and additional ammonia and nitrite are removed by bacteria growing on the sides of the tank and the underside of the polystyrene sheets (i.e., fixed-film nitrification). Finally, water collects in a reservoir (sump) and is returned to the rearing tank. The location of the sump may vary. If elevated hydroponic troughs are used, the sump can be located after the biofilter and water would be pumped up to the troughs and returned by gravity to the fish-rearing tank.
The system can be configured so that a portion of the flow is diverted to a particular treatment unit. For example, a small side-stream flow may go to a hydroponic component after solids are removed, while most of the water passes through a biofilter and returns to the rearing tank.
The biofilter and hydroponic components can be combined by using plant support media such as gravel or sand that also functions as biofilter media. Raft hydroponics, which consists of floating sheets of polystyrene and net pots for plant support, can also provide sufficient biofiltration if the plant production area is large enough. Combining biofiltration with hydroponics is a desirable goal because eliminating the expense of a separate biofilter is one of the main advantages of aquaponics. An alternative design combines solids removal, biofiltration and hydroponics in one unit. The hydroponic support media (pea gravel or coarse sand) captures solids and provides surface area for fixed-film nitrification, although with this design it is important not to overload the unit with suspended solids.
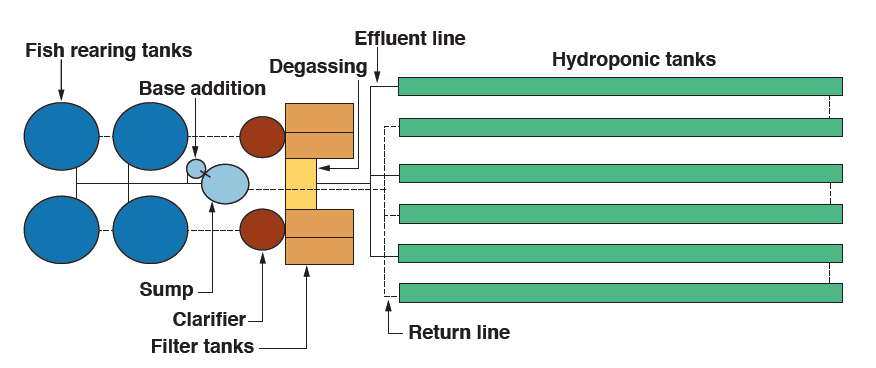
As an example, Figures 3 and 4 show the commercial-scale aquaponic system that has been developed at the University of the Virgin Islands (UVI). It employs raft hydroponics.
Figure 3. Layout of UVI aquaponic system with tank dimensions and pipe sizes (not to scale).
Tank dimensions
- Rearing tanks: Diameter: 10 ft, Height: 4 ft, Water volume: 2,060 gal each
- Clarifiers: Diameter: 6 ft, Height of cylinder: 4 ft, Depth of cone: 3.6 ft, Slope: 45º, Water volume: 1,000 gal
- Filter and degassing tanks: Length: 6 ft, Width: 2.5 ft, Depth: 2 ft, Water volume: 185 gal
- Hydroponic tanks: Length: 100 ft, Width: 4 ft, Depth: 16 in, Water volume: 3,000 gal,
- Growing area: 2,304 ft2
- Sump: Diameter: 4 ft, Height: 3 ft, Water volume: 160 gal
- Base addition tank: Diameter: 2 ft, Height: 3 ft, Water volume: 50 gal
- Total system water volume: 29,375 gal
- Flow rate: 100 GPM
- Water pump: 1⁄2 hp
- Blowers: 11⁄2 hp (fish) and 1 hp (plants)
- Total land area: 1⁄8 acre
Pipe sizes
- Pump to rearing tanks: 3 in
- Rearing tanks to clarifier: 4 in
- Clarifiers to filter tanks: 4 in
- Between filter tanks: 6 in
- Filter tank to degassing tank: 4 in
- Degassing to hydroponic tanks: 6 in
- Between hydroponic tanks: 6 in
- Hydroponic tanks to sump: 6 in
- Sump to pump: 3 in
- Pipe to base addition tank 0.75 in
- Base addition tank to sump: 1.25 in
Figure 4. An early model of the UVI aquaponic system in St. Croix showing the staggered production of leaf lettuce in six raft hydroponic tanks.
Fish Production
Tilapia is the fish species most commonly cultured in aquaponic systems. Although some aquaponic systems have used channel catfish, largemouth bass, crappies, rainbow trout, pacu, common carp, koi carp, goldfish, Asian sea bass (barramundi) and Murray cod, most commercial systems are used to raise tilapia. Most freshwater species, which can tolerate crowding, will do well in aquaponic systems (including ornamental fish). One species reported to perform poorly is hybrid striped bass. They cannot tolerate high levels of potassium, which is often supplemented to promote plant growth.
To recover the high capital cost and operating expenses of aquaponic systems and earn a profit, both the fishrearing and the hydroponic vegetable components must be operated continuously near maximum production capacity. The maximum biomass of fish a system can support without restricting fish growth is called the critical standing crop. Operating a system near its critical standing crop uses space efficiently, maximizes production and reduces variation in the daily feed input to the system, an important factor in sizing the hydroponic component. There are three stocking methods that can maintain fish biomass near the critical standing crop: sequential rearing, stock splitting and multiple rearing units.
Sequential rearing
Sequential rearing involves the culture of several age groups (multiple cohorts) of fish in the same rearing tank. When one age group reaches marketable size, it is selectively harvested with nets and a grading system, and an equal number of fingerlings are immediately restocked in the same tank. There are three problems with this system: 1) the periodic harvests stress the remaining fish and could trigger disease outbreaks; 2) stunted fish avoid capture and accumulate in the system, wasting space and feed; and 3) it is difficult to maintain accurate stock records over time, which leads to a high degree of management uncertainty and unpredictable harvests.
Stock splitting
Stock splitting involves stocking very high densities of fingerlings and periodically splitting the population in half as the critical standing crop of the rearing tank is reached. This method avoids the carryover problem of stunted fish and improves stock inventory. However, the moves can be very stressful on the fish unless some sort of “swimway” is installed to connect all the rearing tanks. The fish can be herded into the swimway through a hatch in the wall of a rearing tank and maneuvered into another rearing tank by movable screens. With swimways, dividing the populations in half involves some guesswork because the fish cannot be weighed or counted. An alternative method is to crowd the fish with screens and pump them to another tank with a fish pump.
Multiple rearing units
With multiple rearing units, the entire population is moved to larger rearing tanks when the critical standing crop of the initial rearing tank is reached. The fish are either herded through a hatch between adjoining tanks or into “swimways” connecting distant tanks. Multiple rearing units usually come in modules of two to four tanks and are connected to a common filtration system. After the largest tank is harvested, all of the remaining groups of fish are moved to the next largest tank and the smallest tank is restocked with fingerlings. A variation of the multiple rearing unit concept is the division of a long raceway into compartments with movable screens. As the fish grow, their compartment is increased in size and moved closer to one end of the raceway where they will eventually be harvested. These should be cross-flow raceways, with influent water entering the raceway through a series of ports down one side of the raceway and effluent water leaving the raceway through a series of drains down the other side. This system ensures that water is uniformly high quality throughout the length of the raceway.
Another variation is the use of several tanks of the same size. Each rearing tank
contains a different age group of fish, but they are not moved during the production
cycle. This system does not use space efficiently in the early stages of growth, but
the fish are never disturbed and the labor involved in moving the fish is eliminated.
A system of four multiple rearing tanks has been used successfully with tilapia in
the UVI commercial scale aquaponic system (Figures 3 and 5). Production is staggered
so one of the rearing tanks is harvested every six weeks. At harvest, the rearing
tank is drained and all of the fish are removed. The rearing tank is then refilled
with the same water and immediately restocked with fingerlings for a 24-week production
cycle. Each circular rearing tank has a water volume of 2,060 gallons and is heavily
aerated with 22 air diffusers. The flow rate to all four tanks is 100 gallons/minute,
but the flow rate to individual tanks is apportioned so that tanks receive a higher
flow rate as the fish grow. The average rearing tank retention time is 82 minutes.
Annual production has been 9,152 pounds (4.16 metric tons) for Nile tilapia and 10,516
pounds (4.78 metric tons) for red tilapia (Table 1). However, production can be increased
to 11,000 pounds (5 metric tons) with close observation of the ad libitum feeding
response.
Figure 5. The UVI aquaponic system at the New Jersey EcoComplex at Rutgers University. Effluent from four tilapiarearing tanks circulates through eight raft hydroponic tanks, producing tomatoes and other crops.
Table 1. Average production values for male mono-sex Nile and red tilapia in the UVI aquaponic system. Nile tilapia are stocked at 0.29 fish/gallon (77 fish per cubed meter) and red tilapia are stocked at 0.58 fish/gallon (154 fish per cubed meter).
| Tilapia | Harvest weight per tank (lbs) | Harvest weight per unit volume (lb/gal) | Initial weight (g/fish) |
|---|---|---|---|
| Nile | 1,056 (480 kg) | 0.51 (61.5 kg/m3) | 79.2 |
| Red | 1,212 (551 kg) | 0.59 (70.7 kg/m3) | 58.8 |
| Tilapia | Final weight (g/fish) |
Growth rate (g/day) |
Survival (%) |
FCR |
|---|---|---|---|---|
| Nile | 813.8 | 4.4 | 98.3 | 1.7 |
| Red | 512.5 | 2.7 | 89.9 | 1.8 |
In general, the critical standing crop in aquaponic systems should not exceed 0.50 pound/gallon. This density will promote fast growth and efficient feed conversion and reduce crowding stress that may lead to disease outbreaks. Pure oxygen is generally not needed to maintain this density.
The logistics of working with both fish and plants can be challenging. In the UVI system, one rearing tank is stocked every six weeks. Therefore, it takes 18 weeks to fully stock the system. If multiple units are used, fish may be stocked and harvested as frequently as once a week. Similarly, staggered crop production requires frequent seeding, transplanting, harvesting and marketing. Therefore, the goal of the design process is to reduce labor wherever possible and make operations as simple as possible. For example, purchasing four fish-rearing tanks adds extra expense. One larger tank could be purchased instead and partially harvested and partially restocked every six weeks. However, this operation requires additional labor, which is a recurring cost and makes management more complex. In the long run, having several smaller tanks in which the fish are not disturbed until harvest (hence, less mortality and better growth) will be more cost effective.
Solids
Most of the fecal waste fish generate should be removed from the waste stream before it enters the hydroponic tanks. Other sources of particulate waste are uneaten feed and organisms (e.g., bacteria, fungi and algae) that grow in the system. If this organic matter accumulates in the system, it will depress dissolved oxygen (DO) levels as it decays and produce carbon dioxide and ammonia. If deep deposits of sludge form, they will decompose anaerobically (without oxygen) and produce methane and hydrogen sulfide, which are very toxic to fish.
Suspended solids have special significance in aquaponic systems. Suspended solids entering the hydroponic component may accumulate on plant roots and create anaerobic zones that prevent nutrient uptake by active transport, a process that requires oxygen. However, some accumulation of solids may be beneficial. As solids are decomposed by microorganisms, inorganic nutrients essential to plant growth are released to the water, a process known as mineralization. Mineralization supplies several essential nutrients. Without sufficient solids for mineralization, more nutrient supplementation is required, which increases the operating expense and management complexity of the system. However, it may be possible to minimize or eliminate the need for nutrient supplementation if fish stocking and feeding rates are increased relative to plants. Another benefit of solids is that the microorganisms that decompose them are antagonistic to plant root pathogens and help maintain healthy root growth.
SRAC Publication No. 453 (“Recirculating Aquaculture Tank Production Systems: A Review
of Component Options”) describes some of the common devices used to remove solids
from recirculating systems. These include settling basins, tube or plate separators,
the combination particle trap and sludge separator, centrifugal separators, microscreen
filters and bead filters. Sedimentation devices (e.g., settling basins, tube or plate
separators) primarily remove settleable solids (>100 microns), while filtration devices
(e.g., microscreen filters, bead filters) remove settleable and suspended solids.
Solids removal devices vary in regard to efficiency, solids retention time, effluent
characteristics (both solid waste and treated water) and water consumption rate.
Sand and gravel hydroponic substrates can remove solid waste from system water. Solids
remain in the system to provide nutrients to plants through mineralization. With the
high potential of sand and gravel media to clog, bed tillage or periodic media replacement
may be required. The use of sand is becoming less common, but one popular aquaponic
system uses small beds (8 feet by 4 feet) containing pea gravel ranging from 1⁄8-
to 1⁄4-inch in diameter. The hydroponic beds are flooded several times daily with
system water and then allowed to drain completely, and the water returned to the rearing
tank. During the draining phase, air is brought into the gravel. The high oxygen content
of air (compared to water) speeds the decomposition of organic matter in the gravel.
The beds are inoculated with red worms (Eisenia foetida), which improve bed aeration
and assimilate organic matter.
Solids Removal
The most appropriate device for solids removal in a particular system depends primarily on the organic loading rate (daily feed input and feces production) and secondarily on the plant growing area. For example, if large numbers of fish (high organic loading) are raised relative to the plant growing area, a highly efficient solids removal device, such as a microscreen drum filter, is desirable. Microscreen drum filters capture fine organic particles, which are retained by the screen for only a few minutes before backwashing removes them from the system. In this system, the dissolved nutrients excreted directly by the fish or produced by mineralization of very fine particles and dissolved organic matter may be sufficient for the size of the plant growing area. If small amounts of fish (low organic loading) are raised relative to the plant growing area, then solids removal may be unnecessary, as more mineralization is needed to produce sufficient nutrients for the plants. However, unstabilized solids (solids that have not undergone microbial decomposition) should not be allowed to accumulate on the tank bottom and form anaerobic zones. A reciprocating pea gravel filter (subject to flood and drain cycles), in which incoming water is spread evenly over the entire bed surface, may be the most appropriate device in this situation because solids are evenly distributed in the gravel and exposed to high oxygen levels (21 percent in air as compared to 0.0005 to 0.0007 percent in fish culture water) on the drain cycle. This enhances microbial activity and increases the mineralization rate.
UVI’s commercial-scale aquaponic system relies on two cylindro-conical clarifiers to remove settleable solids. The fiberglass clarifiers have a volume of 1,000 gallons each. The cylindrical portion of the clarifier is situated above ground and has a central baffle that is perpendicular to the incoming water flow (Figure 6). The lower conical portion has a 45-degree slope and is buried below ground. A drain pipe is connected to the apex of the cone. The drain pipe rises vertically out of the ground to the middle of the cylinder and is fitted with a ball valve. Rearing tank effluent enters the clarifier just below the water surface. The incoming water is deflected upward by a 45-degree pipe elbow to dissipate the current. As water flows under the baffle, turbulence diminishes and solids settle on the sides of the cone. The solids accumulate there and form a thick mat that eventually rises to the surface of the clarifier. To prevent this, approximately 30 male tilapia fingerlings are required to graze on the clarifier walls and consolidate solids at the base of the cone. Solids are removed from the clarifier three times daily. Hydrostatic pressure forces solids through the drain line when the ball valve is opened. A second, smaller baffle keeps floating solids from being discharged to the filter tanks.
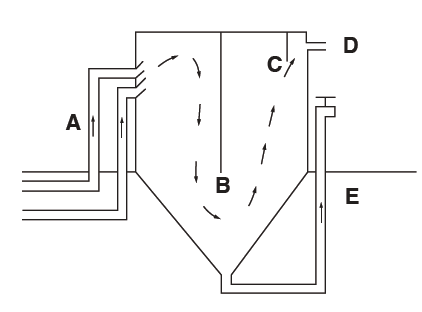
Figure 6. Cross-sectional view (not to scale) of UVI clarifier showing drain lines from two fish rearing tanks (A) central baffle, (B) and discharge baffle, (C) outlet to filter tanks, (D) sludge drain line and (E) direction of water flow (arrows).
The fingerlings serve another purpose. They swim into and through the drain lines and keep them clean. Without tilapia, the 4-inch drain lines would have to be manually cleaned nearly every day because of bacterial growth in the drain lines, which constricts water flow. A cylindrical screen attached to the rearing tank drain keeps fingerlings from entering the rearing tank.
The cylindro-conical clarifier removes approximately 50 percent of the total particulate solids produced by the system and primarily removes large settleable solids. Although fingerlings are needed for effective clarifier performance, their grazing and swimming activities are also counterproductive in that they resuspend some solids, which exit through the clarifier outlet. As fingerlings become larger (>200 g), clarifier performance diminishes. Therefore, clarifier fish must be replaced with small fingerlings (50 g) periodically (once every four months).
With clarification as the sole method of solids removal, large quantities of solids would be discharged to the hydroponic component. Therefore, another treatment stage is needed to remove re-suspended and fine solids. In the UVI system, two rectangular tanks, each with a volume of 185 gallons, are filled with orchard/bird netting and installed after each of the two clarifiers (Figure 7). Effluent from each clarifier flows through a set of two filter tanks in series. Orchard netting is effective in removing fine solids. The filter tanks remove the remaining 50 percent of total particulate solids.
Figure 7. Components of the UVI aquaponic system at the New Jersey EcoComplex at Rutgers University.
The orchard netting is cleaned once or twice each week. Before cleaning, a small sump pump is used to carefully return the filter tank water to the rearing tanks without dislodging the solids. This process conserves water and nutrients. The netting is cleaned with a high-pressure water spray and the sludge is discharged to lined holding ponds.
Effluent from the UVI rearing tanks is highly enriched with dissolved organic matter, which stimulates the growth of filamentous bacteria in the drain line, clarifier and screen tank. The bacteria appear as translucent, gelatinous, light tan filaments. Tilapia consume the bacteria and control its growth in the drain line and clarifier, but bacteria do accumulate in the filter tanks. Without the filter tanks, the bacteria would overgrow plant roots. The bacteria do not appear to be pathogenic, but they do interfere with the uptake of dissolved oxygen, water and nutrients, thereby affecting plant growth. The feeding rate to the system and the flow rate from the rearing tank determine the extent to which filamentous bacteria grow, but they can be contained by providing a sufficient area of orchard netting, either by adjusting screen tank size or using multiple screen tanks. In systems with lower organic loading rates (i.e., feeding rates) or lower water temperature (hence, less biological activity), filamentous bacteria diminish and are not a problem.
The organic matter that accumulates on the orchard netting between cleanings forms a thick sludge. Anaerobic conditions develop in the sludge, which leads to the formation of gases such as hydrogen sulfide, methane and nitrogen. Therefore, a degassing tank is used in the UVI system to receive the effluent from the filter tanks (Figure 7). A number of air diffusers vent the gasses into the atmosphere before the culture water reaches the hydroponic plants. The degassing tank has an internal standpipe well that splits the water flow into three sets of hydroponic tanks.
Solids discharged from aquaponic systems must be disposed of appropriately. There are several methods for effluent treatment and disposal. Effluent can be stored in aerated ponds and applied as relatively dilute sludge to land after the organic matter in it has stabilized. This method is advantageous in dry areas where sludge can be used to irrigate and fertilize field crops. The solid fraction of sludge can be separated from water and used with other waste products from the system (vegetable matter) to form compost. Urban facilities might have to discharge solid waste into sewer lines for treatment and disposal at the municipal wastewater treatment plant.
Biofiltration
A major concern in aquaponic systems is the removal of ammonia, a metabolic waste product excreted through the gills of fish. Ammonia will accumulate and reach toxic levels unless it is removed by the process of nitrification (referred to more generally as biofiltration), in which ammonia is oxidized first to nitrite, which is toxic, and then to nitrate, which is relatively non-toxic. Two groups of naturally occurring bacteria (Nitrosomonas and Nitrobacter) mediate this two-step process. Nitrifying bacteria grow as a film (referred to as biofilm) on the surface of inert material or they adhere to organic particles. Biofilters contain media with large surface areas for the growth of nitrifying bacteria. Aquaponic systems have used biofilters with sand, gravel, shells or various plastic media as substrate. Biofilters perform optimally at a temperature range of 77 to 86 F, a pH range of 7.0 to 9.0, saturated DO, low BOD (<20 milligrams per liter) and total alkalinity of 100 milligrams per liter or more. Nitrification is an acid-producing process. Therefore, an alkaline base must be added frequently, depending on feeding rate, to maintain relatively stable pH values. Some method of removing dead biofilm is necessary to prevent media clogging, short circuiting of water flow, decreasing DO values and declining biofilter performance. A discussion of nitrification principles and a description of various biofilter designs and operating procedures are given in SRAC Publication Nos. 451, 452 and 453.
Four major biofilter options (rotating biological contactors, expandable media filters, fluidized bed filters and packed tower filters) are discussed in SRAC Publication No. 453. If a separate biofilter is required or if a combined biofilter (biofiltration and hydroponic substrate) is used, the standard equations used to size biofilters may not apply to aquaponic systems, as additional surface area is provided by plant roots and a considerable amount of ammonia is taken up by plants. However, the contribution of various hydroponic subsystem designs and plant species to water treatment in aquaponic systems has not been studied. Therefore, aquaponic system biofilters should be sized fairly close to the recommendations for recirculating systems.
Nitrification efficiency is affected by pH. The optimum pH range for nitrification
is 7.0 to 9.0, although most studies indicate that nitrification efficiency is greater
at the higher end of this range (high 8s). Most hydroponic plants grow best at a pH
of 5.8 to 6.2. The acceptable range for hydroponic systems is 5.5 to 6.5. The pH of
a solution affects the solubility of nutrients, especially trace metals. Essential
nutrients such as iron, manganese, copper, zinc and boron are less available to plants
at a pH higher than 7.0, while the solubility of phosphorus, calcium, magnesium and
molybdenum sharply decreases at a pH lower than 6.0. Compromise between nitrification
and nutrient availability is reached in aquaponic systems by maintaining pH close
to 7.0.
Nitrification is most efficient when water is saturated with DO. The UVI commercial-scale
system maintains DO levels near 80 percent saturation (6 to 7 milligrams per liter)
by aerating the hydroponic tanks with numerous small air diffusers (one every 4 feet)
distributed along the long axis of the tanks. Reciprocating (ebb and flow) gravel
systems expose nitrifying bacteria to high atmospheric oxygen levels during the dewatering
phase. The thin film of water that flows through NFT (nutrient film technique) channels
absorbs oxygen by diffusion, but dense plant roots and associated organic matter can
block water flow and create anaerobic zones, which precludes the growth of nitrifying
bacteria and further necessitates the installation of a separate biofilter.
Ideally, aquaponic systems should be designed so that the hydroponic subsystem also serves as the biofilter, which eliminates the capital cost and operational expense of a separate biofilter. Granular hydroponic media such as gravel, sand and perlite provide sufficient substrate for nitrifying bacteria and generally serve as the sole biofilter in some aquaponic systems, although the media has a tendency to clog. If serious clogging occurs from organic matter overloading, gravel and sand filters can actually produce ammonia as organic matter decays, rather than remove it. If this occurs, the gravel or sand must be washed and the system design must be modified by installing a solids removal device before the media, or else the organic loading rate must be decreased by stocking fewer fish and reducing feeding rates.
Raft hydroponics, which consists of channels (with 1 foot of water depth) covered by floating sheets of polystyrene for plant support, also provides sufficient nitrification if solids are removed from the flow before it reaches the hydroponic component. The waste treatment capacity of raft hydroponics is equivalent to a feeding ratio of 180 grams of fish feed per square meter of plant growing area/day. (Note: 1 square meter = 10.76 square feet and 454 grams = 1 lb.) This is equivalent to about 1.2 pounds of feed for each 8-foot x 4-foot sheet of polystyrene foam. After an initial acclimation period of 1 month, it is not necessary to monitor ammonia and nitrite values in the UVI raft system. A significant amount of nitrification occurs on the undersides of the polystyrene sheets, especially in the areas exposed to strong currents above air diffusers where the biofilm is noticeably thicker.
Aquaponic systems using nutrient film technique (NFT) as the hydroponic component may require a separate biofilter. NFT consists of narrow plastic channels for plant support with a film of nutrient solution flowing through them (Figure 8). The water volume and surface area of NFT are considerably smaller than in raft culture because there is just a thin film of water and no substantial side wall area or raft underside surface area for colonization by nitrifying bacteria.
Figure 8. Using nutrient film technique, basil is produced in an aquaponic system at Bioshelters, Inc. in Amherst, Massachusetts.
Hydroponic Subsystems
A number of hydroponic subsystems have been used in aquaponics. Gravel hydroponic subsystems are common in small operations. To ensure adequate aeration of plant roots, gravel beds have been operated in a reciprocating (ebb and flow) mode, where the beds are alternately flooded and drained, or in a nonflooded state, where culture water is applied continuously to the base of the individual plants through smalldiameter plastic tubing. Depending on its composition, gravel can provide some nutrients for plant growth (e.g., calcium is slowly released as the gravel reacts with acid produced during nitrification).
Gravel has several negative aspects. The weight of gravel requires strong support structures. It is subject to clogging with suspended solids, microbial growth and the roots that remain after harvest. The resulting reduction in water circulation, together with the decomposition of organic matter, leads to the formation of anaerobic zones that impair or kill plant roots. The small, plastic tubes used to irrigate gravel are also subject to clogging with biological growth. Moving and cleaning gravel substrate is difficult because of its weight. Planting in gravel is also difficult, and plant stems can be damaged by abrasion in outdoor systems exposed to wind. Gravel retains very little water if drained, so a disruption in flow will lead to the rapid onset of water stress (wilting). The sturdy infrastructure required to support gravel and the potential for clogging limits the size of gravel beds.
One popular gravel-based aquaponic system uses pea gravel in small beds that are irrigated through a distribution system of PVC pipes over the gravel surface. Numerous small holes in the pipes distribute culture water on the flood cycle. The beds are allowed to drain completely between flood cycles. Solids are not removed from the culture water and organic matter accumulates, but the beds are tilled between planting cycles so that some organic matter can be dislodged and discharged.
Sand has been used as hydroponic media in aquaponic systems and is an excellent substrate for plant growth. In an experimental system, sand beds (25 feet long by 5 feet wide by 1.6 feet deep) were constructed on slightly sloped ground covered by polyethylene sheets adjacent to in-ground rearing tanks, with the tank floors sloping to one side. A pump in the deep end of the rearing tank was activated for 30 minutes five times daily to furrow irrigate the adjacent sand bed. The culture water percolated through the sand and returned to the rearing tank. A coarse grade of sand is needed to reduce the potential for clogging over time and some solids should be removed before irrigation.
Perlite is another media that has been used in aquaponic systems. Perlite is placed in shallow aluminum trays (3 inches deep) with a baked enamel finish. The trays vary from 8 inches to 4 feet wide and can be fabricated to any length, with 20 feet the maximum recommended length. At intervals of 20 feet, adjoining trays should be separated by 3 inches or more in elevation so that water drops to the lower tray and becomes re-aerated. A slope of 1 inch per 12 feet is needed for water flow. A small trickle of water enters at the top of the tray, flows through the perlite and keeps it moist, and discharges into a trough at the lower end. Solids must be removed from the water before it enters the perlite tray. Full solids loading will clog the perlite, form short-circuiting channels, create anaerobic zones and lead to non-uniform plant growth. Shallow perlite trays provide minimal area for root growth and are better for smaller plants such as lettuce and herbs.
Nutrient film technique (NFT) has been successfully incorporated into a number of aquaponic systems. NFT consists of many narrow, plastic troughs (4 to 6 inches wide) in which plant roots are exposed to a thin film of water that flows down the troughs, delivering water, nutrients and oxygen to the roots of the plants. The troughs are lightweight, inexpensive and versatile. Troughs can be mounted over rearing tanks to efficiently use vertical greenhouse space. However, this practice is discouraged if it interferes with fish and plant operations such as harvesting. High plant density can be maintained by adjusting the distance between troughs to provide optimum plant spacing during the growing cycle. In aquaponic systems that use NFT, solids must be removed so they do not accumulate and kill roots. With NFT, a disruption in water flow can lead quickly to wilting and death. Water is delivered at one end of the troughs by a PVC manifold with discharge holes above each trough; it is collected at the opposite, down-slope end in an open channel or large PVC pipe. The use of microtubes, which are used in commercial hydroponics, is not recommended because they will clog. The holes should be as large as practical to reduce cleaning frequency.
A floating or raft hydroponic subsystem is ideal for the cultivation of leafy green and other types of vegetables. The UVI system uses three sets of two raft hydroponic tanks that are 100 feet long by 4 feet wide by 16 inches deep and contain 12 inches of water. The channels are lined with low-density polyethylene liners (20 mil thick) and covered by expanded polystyrene sheets (rafts) that are 8 feet long by 4 feet wide by 1.5 inches thick. Net pots are placed in holes in the raft and just touch the water surface. Two-inch net pots are generally used for leafy green plants, while 3- inch net pots are used for larger plants such as tomatoes or okra. Holes of the same size are cut into the polystyrene sheet. A lip at the top of the net pot secures it and keeps it from falling through the hole into the water. Seedlings are nursed in a greenhouse and then placed into net pots. Their roots grow into the culture water while their canopy grows above the raft surface. The system provides maximum exposure of roots to the culture water and avoids clogging. The sheets shield the water from direct sunlight and maintain lower than ambient water temperature, which is a beneficial feature in tropical systems. A disruption in pumping does not affect the plant’s water supply as in gravel, sand and NFT subsystems. The sheets are easily moved along the channel to a harvesting point where they can be lifted out of the water and placed on supports at an elevation that is comfortable for workers (Figure 9).
Figure 9. Leaf lettuce being harvested from a raft hydroponic tank in the UVI aquaponic system in St. Croix.
A disadvantage of rafts in an aquaponic system is that roots are exposed to harmful organisms associated with aquaculture systems. If tilapia fry gain access to the hydroponic tanks, they consume plant roots and severely stunt plant growth, although it is relatively easy to keep fish from entering by placing a fine mesh screen at the entry point of water into the degassing tank. Similarly, blooms of zooplankton, especially ostracods, will consume root hairs and fine roots, retarding plant growth. Other pests are tadpoles and snails, which consume roots and nitrifying bacteria. These problems can be surmounted by increasing water agitation to prevent root colonization by zooplankton and by stocking some carnivorous fish such as red ear sunfish (shellcrackers) in hydroponic tanks to prey on tadpoles and snails.
Sump
Water flows by gravity from gravel, sand and raft hydroponic subsystems to a sump, which is the lowest point in the system. The sump contains a pump or pump inlet that returns the treated culture water to the rearing tanks. If NFT troughs or perlite trays are located above the rearing tanks, the sump would be positioned in front of them so that water could be pumped up to the hydroponic component for gravity return to the rearing tanks. There should be only one pump to circulate water in an aquaponic system.
The sump should be the only tank in the system where the water level decreases as a result of overall water loss from evaporation, transpiration, sludge removal and splashing. An electrical or mechanical valve is used to automatically add replacement water from a storage reservoir or well. Municipal water should not be used unless it is de-chlorinated. Surface water should not be used because it may contain disease organisms. A water meter should be used to record additions. Unusually high water consumption indicates a leak.
The sump is a good location for the addition of base to the system. Soluble base such as potassium hydroxide causes high and toxic pH levels in the sump. However, as water is pumped into the rearing tank, it is diluted and pH decreases to acceptable levels. The UVI system has a separate base addition tank located next to the sump. As water is pumped from the sump to the fish-rearing tanks, a small pipe, tapped into the main water distribution line, delivers a small flow of water to the base addition tank, which is well aerated with one large air diffuser. When base is added to this tank and dissolves, the resulting high pH water slowly flows by gravity into the sump, where it is rapidly diluted and pumped to fishrearing tanks. This system prevents a rapid pH increase in the fish-rearing tank.
Construction Materials
Many materials can be used to construct aquaponic systems. Budget limitations often lead to the selection of inexpensive and questionable materials such as vinyl-lined, steelwalled swimming pools. Plasticizers used in vinyl manufacture are toxic to fish, so these liners must be washed thoroughly or aged with water for several weeks before fish can be added safely to a tank of clean water. After a few growing periods, vinyl liners shrink upon drying, become brittle and crack, while the steel walls gradually rust. Nylon-reinforced, neoprene rubber liners are not recommended either. Tilapia eat holes in rubber liners at the folds as they graze on microorganisms. Moreover, neoprene rubber liners are not impervious to chemicals. If herbicides and soil sterilants are applied under or near rubber liners, these chemicals can diffuse into culture water, accumulate in fish tissue and kill hydroponic vegetables.
Fiberglass is the best construction material for rearing tanks, sumps and filter tanks. Fiberglass tanks are sturdy, durable, non-toxic, movable and easy to plumb. Polyethylene tanks are also very popular for fish rearing and gravel hydroponics because of their low cost. NFT troughs made from extruded polyethylene are specifically designed to prevent the puddling and water stagnation that lead to root death and are preferable to makeshift structures such as PVC pipes. Plastic troughs are commercially available for floating hydroponic subsystems, but they are expensive. A good alternative is the 20-mil polyethylene liners that are placed inside concreteblock or poured-concrete side walls. They are easy to install, relatively inexpensive and durable, with an expected life of 12 to 15 years. A soil floor covered with fine sand will prevent sharp objects from puncturing the liners. Lined hydroponic tanks can be constructed to very large sizes—hundreds of feet long and up to 30 feet wide.
Component Ratios
Aquaponic systems are generally designed to meet the size requirements for solids removal (for those systems requiring solids removal) and biofiltration (if a separate biofilter is used) for the quantity of fish being raised (see SRAC Publication No. 453, “Recirculating Aquaculture Tank Production Systems: A Review of Component Options”). After the size requirements are calculated, it is prudent to add excess capacity as a safety margin. However, if a separate biofilter is used, the hydroponic component is the safety factor because a significant amount of ammonia uptake and nitrification will occur regardless of hydroponic technique.
Another key design criterion is the ratio between the fish-rearing and hydroponic components. The key is the ratio of daily feed input to plant growing area. If the ratio of daily feeding rate to plants is too high, nutrient salts will accumulate rapidly and may reach phytotoxic levels. Higher water exchange rates will be required to prevent excessive nutrient buildup. If the ratio of daily feeding rate to plants is too low, plants will develop nutrient deficiencies and need more nutrient supplementation. Fortunately, hydroponic plants grow well over a wide range of nutrient concentrations.
The optimum ratio of daily fish feed input to plant growing area will maximize plant production while maintaining relatively stable levels of dissolved nutrients. A volume ratio of 1 cubed feet of fish-rearing tank to 2 cubed feet of pea gravel hydroponic media (1⁄8- to 1⁄4-inch in diameter) is recommended for reciprocating (flood and drain) gravel aquaponic systems. This ratio requires that tilapia be raised to a final density of 0.5 pound/gallon and fed appropriately. With the recommended ratio, no solids are removed from the system. The hydroponic beds should be cultivated (stirred up) between crops and inoculated with red worms to help break down and assimilate the organic matter. With this system, nutrient supplementation may not be necessary.
As a general guide for raft aquaponics, a ratio in the range of 60 to 100 grams of fish feed per square meter of plant growing area per day should be used. Ratios within this range have been used successfully in the UVI system for the production of tilapia, lettuce, basil and several other plants. In the UVI system all solids are removed, with a residence time of <1 day for settleable solids (>100 micrometers) removed by a clarifier, and 3 to 7 days for suspended solids removed by an orchard netting filter. The system uses rainwater and requires supplementation for potassium, calcium and iron.
Another factor to consider in determining the optimum feeding rate ratio is the total water volume of the system, which affects nutrient concentrations. In raft hydroponics, approximately 75 percent of the system water volume is in the hydroponic component, whereas gravel beds and NFT troughs contain minor amounts of system water.
Theoretically, in systems producing the same quantity of fish and plants, a daily feeding rate of 100 grams per square meter would produce total nutrient concentrations nearly four times higher in gravel and NFT systems (e.g., 1,600 milliliters per liter) than in raft systems (e.g., 400 milliliters per liter), but total nutrient mass in the systems would be the same. Nutrient concentrations outside acceptable ranges affect plant growth. Therefore, the optimum design ratio varies with the type of hydroponic component. Gravel and NFT systems should have a feeding rate ratio that is approximately 25 percent of the recommended ratio for raft hydroponics.
Other factors in determining the optimum feeding rate ratio are the water exchange rate, nutrient levels in the source water, degree and speed of solids removal, and type of plant being grown. Lower rates of water exchange, higher source-water nutrient levels, incomplete or slow solids removal (resulting in the release of more dissolved nutrients through mineralization), and slow-growing plants would allow a lower feeding rate ratio. Conversely, higher water exchange rates, low source-water nutrient levels, rapid and complete solids removal, and fast-growing plants would allow a higher feeding rate ratio.
The optimum feeding rate ratio is influenced by the plant culture method. With batch culture, all plants in the system are planted and harvested at the same time. During their maximum growth phase, there is a large uptake of nutrients, which requires a higher feeding rate ratio during that period. In practice, however, a higher feeding rate ratio is used throughout the production cycle. With a staggered production system, plants are in different stages of growth, which levels out nutrient uptake rates and allows good production with slightly lower feeding rate ratios.
In properly designed aquaponic systems, the surface area of the hydroponic component is large compared to the surface area of the fish-rearing tank (stocked at commercially relevant densities). The commercial scale unit at UVI has a ratio of 7.3:1. The total plant growing area is 2,304 square feet and the total fish-rearing surface area is 314 square feet.
Plant Growth Requirements
For maximum growth, plants in aquaponic systems require 16 essential nutrients. These are listed below in the order of their concentrations in plant tissue, with carbon and oxygen being the highest. The essential elements are arbitrarily divided into macronutrients, those required in relatively large quantities, and micronutrients, those required in considerably smaller amounts. Three of the macronutrients—carbon (C), oxygen (O) and hydrogen (H)—are supplied by water (H2O) and carbon dioxide gas (CO2). The remaining nutrients are absorbed from the culture water. Other macronutrients include nitrogen (N), potassium (K), calcium (Ca), magnesium (Mg), phosphorus (P) and sulfur (S). The seven micronutrients include chlorine (Cl), iron (Fe), manganese (Mn), boron (B), zinc (Zn), copper (Cu) and molybdenum (Mo). These nutrients must be balanced for optimum plant growth. High levels of one nutrient can influence the bioavailability of others. For example, excessive amounts of potassium may interfere with the uptake of magnesium or calcium, while excessive amounts of either of the latter nutrients may interfere with the uptake of the other two nutrients.
Enriching the air in an unventilated greenhouse with CO2 has dramatically increased crop yields in northern latitudes. Doubling atmospheric CO2 increases agricultural yields by an average of 30 percent. However, the high cost of energy to generate CO2 has discouraged its use. An aquaponic system in a tightly enclosed greenhouse is ideal because CO2 is constantly vented from the culture water.
There is a growing body of evidence that healthy plant development relies on a wide range of organic compounds in the root environment. These compounds, generated by complex biological processes involving microbial decomposition of organic matter, include vitamins, auxins, gibberellins, antibiotics, enzymes, coenzymes, amino acids, organic acids, hormones and other metabolites. Directly absorbed and assimilated by plants, these compounds stimulate growth, enhance yields, increase vitamin and mineral content, improve fruit flavor and hinder the development of pathogens. Various fractions of dissolved organic matter (e.g., humic acid) form organo-metallic complexes with Fe, Mn and Zn, thereby increasing the availability of these micronutrients to plants. Although inorganic nutrients give plants an avenue to survival, plants not only use organic metabolites from the environment, but also need these metabolites to reach their full growth potential.
Maintaining high DO levels in the culture water is extremely important for optimal
plant growth, especially in aquaponic systems with their high organic loads. Hydroponic
plants are subject to intense root respiration and draw large amounts of oxygen from
the surrounding water. If DO is deficient, root respiration decreases. This reduces
water absorption, decreases nutrient uptake, and causes the loss of cell tissue from
roots. The result is reduced plant growth. Low DO levels correspond with high concentrations
of carbon dioxide, a condition that promotes the development of plant root pathogens.
Root respiration, root growth and transpiration are greatest at saturated DO levels.
Climatic factors also are important for hydroponic plant production. Production is
generally best in regions with maximum intensity and daily duration of light. Growth
slows substantially in temperate greenhouses during winter because solar radiation
is low. Supplemental illumination can improve winter production, but is not generally
cost effective unless an inexpensive energy source is available.
Water temperature is far more important than air temperature for hydroponic plant production. The best water temperature for most hydroponic crops is about 75 F. However, water temperature can go as low as the mid-60s for most common garden crops and slightly lower for winter crops such as cabbage, brussels sprouts and broccoli. Maintaining the best water temperature requires heating during the winter in temperate greenhouses and year-round cooling in tropical greenhouses. In addition to evaporative cooling of tropical greenhouses, chillers are often used to cool the nutrient solution. In tropical outdoor systems, complete shading of the fish-rearing and filtration components lowers system water temperature. In raft hydroponics, the polystyrene sheets shield water from direct sunlight and maintain temperatures that are several degrees lower than those in open bodies of water.
Crop varieties may need to be adjusted seasonally for both temperate and tropical aquaponic production. Plants cultured in outdoor aquaponic systems must be protected from strong winds, especially after transplanting when seedlings are fragile and most vulnerable to damage.
Nutrient Dynamics
Dissolved nutrients are measured collectively as total dissolved solids (TDS), expressed as ppm, or as the capacity of the nutrient solution to conduct an electrical current (EC), expressed as millimhos/cm (mmho/cm). In a hydroponic solution, the recommended range for TDS is 1,000 to 1,500 ppm (1.5 to 3.5 mmho/cm). In an aquaponic system, considerably lower levels of TDS (200 to 400 ppm) or EC (0.3 to 0.6 mmho/cm) will produce good results because nutrients are generated continuously. A concern with aquaponic systems is nutrient accumulation. High feeding rates, low water exchange and insufficient plant growing areas can lead to the rapid buildup of dissolved nutrients to potentially phytotoxic levels. Phytotoxicity occurs at TDS concentrations above 2,000 ppm or EC above 3.5 mmho/cm. Because aquaponic systems have variable environmental conditions such as daily feed input, solids retention, mineralization, water exchange, nutrient input from source water or supplementation, and variable nutrient uptake by different plant species, it is difficult to predict the exact level of TDS or EC and how it is changing. Therefore, the culturist should purchase an inexpensive conductivity meter and periodically measure TDS or EC. If dissolved nutrients are steadily increasing and approach 2,000 ppm as TDS or 3.5 mmho/cm as EC, increasing the water exchange rate or reducing the fish stocking rate and feed input will quickly reduce nutrient accumulation. However, because these methods either increase costs (i.e., more water consumed) or lower output (i.e., less fish produced), they are not good long-term solutions. Better but more costly solutions involve removing more solids (i.e., upgrade the solids removal component) or enlarging the plant-growing areas.
The major ions that increase conductivity are nitrate (NO3-), phosphate (PO4 -2), sulfate (SO4 -2), K+, Ca+2 and Mg+2. Levels of NO3 -, PO4 -2 and SO4 -2 are usually sufficient for good plant growth, while levels of K+ and Ca+2 are generally insufficient. Potassium is added to the system in the form of potassium hydroxide (KOH) and Ca is added as calcium hydroxide [Ca(OH)2]. In the UVI commercialscale system, KOH and Ca(OH)4 are added in equal amounts (usually 500 to 1,000 g). The bases are added alternately several times weekly to maintain pH near 7.0. Adding basic compounds of K and Ca serves the dual purpose of supplementing essential nutrients and neutralizing acid. In some systems Mg also may be limiting. Magnesium can be supplemented by using dolomite [CaMg(CO3)2] as the base to adjust pH. The addition of too much Ca can cause phosphorous to precipitate from culture water in the form of dicalcium phosphate [CaHPO4]. Sodium bicarbonate (NaHCO3) should never be added to an aquaponic system for pH control because a high Na+ level in the presence of chloride is toxic to plants. The Na+ concentration in hydroponic nutrient solutions should not exceed 50 milligrams per liter. Higher Na+ levels will interfere with the uptake of K+ and Ca+2. In lettuce, reduced Ca+2 uptake causes tip-burn, resulting in an unmarketable plant. Tip-burn often occurs during the warmer months. Salt (NaCl) is added to fish feed during manufacture. A producer who orders large quantities of feed could request that salt not be added if this does not affect fish health. If Na+ exceeds 50 milligrams per liter and the plants appear to be affected, a partial water exchange (dilution) may be necessary. Rainwater is used in UVI’s systems because the groundwater of semiarid islands generally contains too much salt for aquaponics.
The accumulation of too much nitrate in aquaponic systems is sometimes a concern as fruiting plants set less fruit and produce excess vegetative growth when nitrate levels are high. The filter tanks in the UVI commercial-scale system have a mechanism for controlling nitrate levels through denitrification, the reduction of nitrate ions to nitrogen gas by anaerobic bacteria. Large quantities of organic matter accumulate on the orchard netting between cleanings. Denitrification occurs in anaerobic pockets that develop in the sludge. Water moves through the accumulated sludge, which provides good contact between nitrate ions and denitrifying bacteria. The frequency of cleaning the netting regulates the degree of denitrification. When the netting is cleaned often (e.g., twice per week), sludge accumulation and denitrification are minimized, which leads to an increase in nitrate concentrations. When the netting is cleaned less often (e.g., once per week), sludge accumulation and denitrification are maximized, which leads to a decrease in nitrate levels. Nitrate-nitrogen levels can be regulated within a range of 1 to 100 milligrams per liter or more. High nitrate concentrations promote the growth of leafy green vegetables, while low nitrate concentrations promote fruit development in vegetables such as tomatoes.
The micronutrients Fe+2, Mn+2, Cu+2, B+3 and Mo+6 do not accumulate significantly in aquaponic systems with respect to cumulative feed input. The Fe+2 derived from fish feed is insufficient for hydroponic vegetable production and must be supplemented with chelated Fe+2 so that the concentration of Fe+2 is 2.0 milligrams per liter. Chelated Fe+2 has an organic compound attached to the metal ion to prevent it from precipitating out of solution and making it unavailable to plants. The best chelate is Fe-DTPA because it remains soluble at pH 7.0. Fe-EDTA is commonly used in the hydroponics industry, but it is less stable at pH 7.0 and needs to be replenished frequently. Fe+2 also can be applied in a foliar spray directly to plant leaves. A comparison of Mn+2, B+3 and Mo+6 levels with standard nutrient formulations for lettuce shows that their concentrations in aquaponic systems are several times lower than their initial levels in hydroponic formulations. Deficiency symptoms for Mn+2, B+3 and Mo+6 are not detected in aquaponic systems, so their concentrations appear to be adequate for normal plant growth. Concentrations of Cu+2 are similar in aquaponic systems and hydroponic formulations, while Zn+2 accumulates in aquaponic systems to levels that are four to sixteen times higher than initial levels in hydroponic formulations. Nevertheless, Zn+2 concentrations usually remain within the limit that is safe for fish.
Vegetable Selection
Many types of vegetables have been grown in aquaponic systems. However, the goal is to culture a vegetable that will generate the highest level of income per unit area per unit time. With this criterion, culinary herbs are the best choice. They grow very rapidly and command high market prices. The income from herbs such as basil, cilantro, chives, parsley, portulaca and mint is much higher than that from fruiting crops such as tomatoes, cucumbers, eggplant and okra. For example, in experiments in UVI’s commercial scale system, basil production was 11,000 pounds annually at a value of $110,000, compared to okra production of 6,400 pounds annually at a value of $6,400. Fruiting crops also require longer culture periods (90 days or more) and have more pest problems and diseases. Lettuce is another good crop for aquaponic systems because it can be produced in a short period (3 to 4 weeks in the system) and, as a consequence, has relatively few pest problems. Unlike fruiting crops, a large portion of the harvested biomass is edible. Other suitable crops are Swiss chard, pak choi, Chinese cabbage, collard and watercress. The cultivation of flowers has potential in aquaponic systems. Good results have been obtained with marigold and zinnia in UVI’s aquaponic system. Traditional medicinal plants and plants used for the extraction of modern pharmaceuticals have not been cultivated in aquaponic systems, but there may be potential for growing some of these plants. All plant production has to be coupled to the producer’s ability to market the final product.
Crop Production Systems
There are three strategies for producing vegetable crops in the hydroponic component. These are staggered cropping, batch cropping and intercropping. A staggered crop production system is one in which groups of plants in different stages of growth are cultivated simultaneously. This allows produce to be harvested regularly and keeps the uptake of nutrients from the culture water relatively constant. This system is most effective where crops can be grown continuously, as in the tropics, subtropics, or temperate greenhouses with environmental control. At UVI, the production of leaf lettuce is staggered so that a crop can be harvested weekly on the same day, which facilitates marketing arrangements. Bibb lettuce reaches market size three weeks after transplanting. Therefore, three growth stages of Bibb lettuce are cultivated simultaneously, and one-third of the crop is harvested weekly. Red leaf lettuce and green leaf lettuce require four weeks to reach marketable size. The cultivation of four growth stages of these lettuce varieties allows one-fourth of the crop to be harvested weekly. In three years of continuous operation, UVI has harvested 148 crops of lettuce, which demonstrates the system’s sustainability. Leafy green vegetables, herbs and other crops with short production periods are well suited for continuous, staggered production systems.
A batch cropping system is more appropriate for crops that are grown seasonally or have long growing periods (>3 months), such as tomatoes and cucumbers. Various intercropping systems can be used in conjunction with batch cropping. For example, if lettuce is intercropped with tomatoes and cucumbers, one crop of lettuce can be harvested before the tomato plant canopy begins to limit light.
Pest and Disease Control
Pesticides should not be used to control insects on aquaponic plant crops. Even pesticides that are registered would pose a threat to fish and would not be permitted in a fish culture system. Similarly, therapeutants for treating fish parasites and diseases should not be used because vegetables may absorb and concentrate them. The common practice of adding salt to treat fish diseases or reduce nitrite toxicity is detrimental to plant crops. Nonchemical methods of integrated pest management must be used. These include biological control (resistant cultivars, predators, pathogens, antagonistic organisms), physical barriers, traps, and manipulation of the physical environment. There are more opportunities to use biological control methods in enclosed greenhouse environments than in exterior installations. Parasitic wasps and ladybugs can be used to control white flies and aphids. In UVI’s systems, caterpillars are effectively controlled by twice weekly spraying with Bacillus thuringiensis, a bacterial pathogen that is specific to caterpillars. Fungal root pathogens (Pythium), which are encountered in summer at UVI and reduce production, dissipate in winter in response to lower water temperature.
The prohibition on the use of pesticides makes crop production in aquaponic systems more difficult. However, this restriction ensures that crops from aquaponic systems will be raised in an environmentally sound manner and be free of pesticide residues. A major advantage of aquaponic systems is that crops are less susceptible to attack from soilborne diseases. Plants grown in aquaponic systems may be more resistant to diseases that affect plants grown in standard hydroponics. This resistance may be due to the presence of some organic matter in the culture water that creates a stable growing environment with a wide diversity of microorganisms, some of which may be antagonistic to plant root pathogens (Figure 10).
Figure 10. Healthy roots of Italian parsley cultured on rafts in a UVI aquaponic system at the Crop Diversification Center South in Alberta, Canada.
Approaches to System Design
There are several ways to design an aquaponic system. The simplest approach is to duplicate a standard system or scale a standard system down or up, keeping the components proportional. Changing aspects of the standard design is not recommended because changes often lead to unintended consequences. However, the design process often starts with a production goal for either fish or plants. In those cases there are some guidelines that can be followed.
Use an aquaponic system that is already designed. The easiest approach is to use a system design that has been tested and is in common use with a good track record. It is early in the development of aquaponics, but standard designs will emerge. The UVI system has been well documented and is being studied or used commercially in several locations, but there are other systems with potential. Standard designs will include specifications for layout, tank sizes, pipe sizes, pipe placement, pumping rates, aeration rates, infrastructure needs, etc. There will be operation manuals and projected production levels and budgets for various crops. Using a standard design will reduce risk.
Design for available space. If a limited amount of space is available, as in an existing greenhouse, then that space will define the size of the aquaponic system. A standard design can be scaled down to fit the space. If a scaled-down tank or pipe size falls between commercially available sizes, it is best to select the larger size. However, the water flow rate should equal the scaled-down rate for best results. The desired flow rate can be obtained by buying a higher capacity pump and installing a bypass line and valve, which circulates a portion of the flow back to the sump and allows the desired flow rate to go from the pump to the next stage of the system. If more space is available than the standard design requires, then the system could be scaled up within limitations or more than one scaled-down system could be installed.
Design for fish production. If the primary objective is to produce a certain amount of fish annually, the first step in the design process will be to determine the number of systems required, the number of rearing tanks required per system, and the optimum rearing tank size. The number of harvests will have to be calculated based on the length of the culture period. Assume that the final density is 0.5 pound/gallon for an aerated system. Take the annual production per system and multiply it by the estimated feed conversion ratio (the pounds of feed required to produce 1 pound of fish). Convert the pounds of annual feed consumption to grams (454 grams per pound) and divide by 365 days to obtain the average daily feeding rate. Divide the average daily feeding rate by the desired feeding rate ratio, which ranges from 60 to 100 grams per square meter per day for raft culture, to determine the required plant production area. For other systems such as NFT, the feeding rate ratio should be decreased in proportion to the water volume reduction of the system as discussed in the component ratio section. Use a ratio near the low end of the range for small plants such as Bibb lettuce and a ratio near the high end of the range for larger plants such as Chinese cabbage or romaine lettuce. The solids removal component, water pump and blowers should be sized accordingly
Sample problem:
This example illustrates only the main calculations, which are simplified (e.g., mortality is not considered) for the sake of clarity. Assume that you have a market for 500 pounds of live tilapia per week in your city and that you want to raise lettuce with the tilapia because there is a good market for green leaf lettuce in your area. The key questions are: How many UVI aquaponic systems do you need to harvest 500 pounds of tilapia weekly? How large should the rearing tanks be? What is the appropriate number and size of hydroponic tanks? What would the weekly lettuce harvest be?
- Each UVI system contains four fish-rearing tanks (Figure 3). Fish production is staggered so that one fish tank is harvested every 6 weeks. The total growing period per tank is 24 weeks. If 500 pounds of fish are required weekly, six production systems (24 fish-rearing tanks) are needed.
- Aquaponic systems are designed to achieve a final density of 0.5 pound per gallon. Therefore, the water volume of the rearing tanks is 1,000 gallons.
- In 52 weeks, there will be 8.7 harvests (52 ÷ 6 = 8.7) per system. Annual production for the system, therefore, is 4,350 pounds (500 pounds per harvest × 8.7 harvests).
- The usual feed conversion ratio is 1.7. Therefore, annual feed input to the system is 7,395 pounds (4,350 pounds × 1.7 = 7,395 pounds).
- The average daily feed input is 20.3 pounds (7,395 pounds per year ÷ 365 days = 20.3 pounds).
- The average daily feed input converted to grams is 9,216 grams (20.3 pounds × 454 grams per pound = 9216 grams).
- The optimum feeding rate ratio for raft aquaponics ranges from 60 to 100 grams per square meter per day. Select 80 grams per square meter per day as the design ratio. Therefore, the required lettuce growing area is 115.2 square meter (9,216 grams per day ÷ 80 grams per square meter per day =115.2 square meter).
- The growing area in square feet is 1,240 (115.2 square meter × 10.76 square feet per square meter = 1,240 square feet).
- Select a hydroponic tank width of 4 feet. The total length of the hydroponic tanks is 310 feet (1,240 square feet ÷ 4 feet = 310 feet).
- Select four hydroponic tanks. They are 77.5 feet long (310 feet ÷ 4 = 77.5 feet). They are rounded up to 80 feet in length, which is a practical length for a standard greenhouse and allows the use of ten 8-foot sheets of polystyrene per hydroponic tank.
- Green leaf lettuce produces good results with plant spacing of 48 plants per sheet (16 per square meter). The plants require a four-week growth period. With staggered production, one hydroponic tank is harvested weekly. Each hydroponic tank with ten polystyrene sheets produces 480 plants. With six aquaponic production systems 2,880 plants are harvested weekly.
In summary, the weekly production of 500 pounds of tilapia results in the production of 2,880 green leaf lettuce plants (120 cases). Six aquaponic systems, each with four 1,000-gallon rearing tanks (water volume), are required. Each system will have four raft hydroponic tanks that are 80 feet long by 4 feet wide.
Design for plant production. If the primary objective is to produce a certain quantity of plant crops annually, the first step in the design process will be to determine the area required for plant production. The area needed will be based on plant spacing, length of the production cycle, number of crops per year or growing season, and the estimated yield per unit area and per crop cycle. Select the desired feeding rate ratio and multiple by the total area to obtain the average daily feeding rate required. Multiply the average daily feeding rate by 365 days to determine annual feed consumption. Estimate the feed conversion ratio (FCR) for the fish species that will be cultured. Convert FCR to feed conversion efficiency. For example, if FCR is 1.7:1, then the feed conversion efficiency is 1 divided by 1.7 or 0.59. Multiply the annual feed consumption by the feed conversion efficiency to determine net annual fish yield. Estimate the average fish weight at harvest and subtract the anticipated average fingerling weight at stocking. Divide this number into the net annual yield to determine the total number of fish produced annually. Multiply the total number of fish produced annually by the estimated harvest weight to determine total annual fish production. Divide total annual fish production by the number of production cycles per year. Take this number and divide by 0.5 pound/gallon to determine the total volume that must be devoted to fish production. The required water volume can be partitioned among multiple systems and multiple tanks per system with the goal of creating a practical system size and tank array. Divide the desired individual fish weight at harvest by 0.5 pound/gallon to determine the volume of water (in gallons) required per fish. Divide the number of gallons required per fish by the water volume of the rearing tank to determine the fish stocking rate. Increase this number by 5 to 10 percent to allow for expected mortality during the production cycle. The solids removal component, water pump and blowers should be sized accordingly.
Sample problem:
Assume that there is a market for 1,000 Bibb lettuce plants weekly in your city. These plants will be sold individually in clear, plastic, clamshell containers. A portion of the root mass will be left intact to extend self life. Bibb lettuce transplants are cultured in a UVI raft system for three weeks at a density of 29.3 plants per square meter. Assume that tilapia will be grown in this system. The key questions are: How large should the plant growing area be? What will be the annual production of tilapia? How large should the fish-rearing tanks be?
- Bibb lettuce production will be staggered so that 1,000 plants can be harvested weekly. Therefore, with a three-week growing period, the system must accommodate the culture of 3,000 plants.
- At a density of 29.3 plants per square meter, the total plant growing area will be 102.3 square meter (3,000 plants ÷ 29.3/square meter = 102.3 square meter). This area is equal to 1,100 square feet (102.3 square meter × 10.76 square feet per square meter = 1,100 square feet).
- Select a hydroponic tank width of 8 feet. The total hydroponic tank length will be 137.5 feet (1,100 square feet per 8 feet = 137.5 feet).
- Multiples of two raft hydroponic tanks are required for the UVI system. In this case only two hydroponic tanks are required. Therefore, the minimum length of each hydroponic tank will be 68.75 feet (137.5 feet ÷ 2 = 68.75 feet). Since polystyrene sheets come in 8-foot lengths, the total number of sheets per hydroponic tank will be 8.59 sheets (68.75 feet ÷ 8 feet per sheet = 8.59 sheets). To avoid wasting material, round up to nine sheets. Therefore, the hydroponic tanks will be 72 feet long (9 sheets × 8 feet per sheet = 72 feet).
- The total plant growing area will then be 1,152 square feet (72 feet × 8 feet per tank × 2 tanks = 1,152 square feet). This is equal to 107 square meters (1,152 square feet ÷ 10.76 square feet per square meter).
- At a planting density of 29.3 plants per square meter, a total of 3,135 plants will be cultured in the system. The extra plants will provide a safety margin against mortality and plants that do not meet marketing standards.
- Assume that a feeding rate of 60 grams per square meter per day provides sufficient nutrients for good plant growth. Therefore, daily feed input to the system will be 6,420 grams (60 grams per square meter per day × 107 square meters = 6,420 grams). This is equal to 14.1 pounds of feed (6,420 grams ÷ 454 grams per pound = 14.1 pounds).
- Annual feed input to the system will be 5,146 pounds (14.1 pounds per day × 365 days = 5,146 pounds)
- Assume the feeding conversion ratio is 1.7. Therefore, the feed conversion efficiency is 0.59 (1 pounds of gain ÷ 1.7 pounds of feed = 0.59).
- The total annual fish production gain will be 3,036 pounds (5,146 pounds × 0.59 feed conversion efficiency = 3,036 pounds).
- Assume that the desired harvest weight of the fish will be 500 grams (1.1 pounds) and that 50-gram (01.1-pounds) fingerling will be stocked. Therefore, individual fish will gain 450 grams (500 grams harvest weight – 50 grams stocking weight = 450 grams). The weight gain per fish will be approximately 1 pound (454 grams).
- The total number of fish harvested will be 3,036 (3,036 pounds of total gain ÷ 1 pound of gain per fish = 3,036 fish).
- Total annual production will be 3,340 pounds (3,036 fish × 1.1 pounds per fish = 3,340 pounds) when the initial stocking weight is considered.
- If there are four fish-rearing tanks and one tank is harvested every six weeks, there will be 8.7 harvests per year (52 weeks ÷ 6 weeks = 8.7).
- Each harvest will be 384 pounds (3,340 pounds per year ÷ 8.7 harvests per year = 384 pounds per harvest).
- Final harvest density should not exceed 0.5 pound/gallon. Therefore, the water volume of each rearing tank should be 768 gallons (384 pound ÷ 0.5 pounds per gallon = 768 gallon). The tank should be larger to provide a 6-inch freeboard (space between the top edge of the tank and the water levels).
- Each fish requires 2.2 gallons of water (1.1 pounds ÷ 0.5 pound of fish/gal = 2.2 gallons per fish).
- The stocking rate is 349 fish per tank (768 gallons ÷ 2.2 gallons per fish = 349 fish).
- To account for calculated mortality, the stocking rate (349 fish per tank) should be increased by 35 fish (349 fish × 0.10 = 34.9) to attain an actual stocking of 384 fish per tank.
In summary, two hydroponic tanks (each 72 feet long by 8 feet wide) will be required to produce 1,000 Bibb lettuce plants per week. Four fish-rearing tanks with a water volume of 768 gallons per tank will be required. The stocking rate will be 384 fish per tank. Approximately 384 pounds of tilapia will be harvested every six weeks, and annual tilapia production will be 3,340 pounds.
Economics
The economics of aquaponic systems depends on specific site conditions and markets. It would be inaccurate to make sweeping generalizations because material costs, construction costs, operating costs and market prices vary by location. For example, an outdoor tropical system would be less expensive to construct and operate than a controlled-environment greenhouse system in a temperate climate. Nevertheless, the economic potential of aquaponic systems looks promising based on studies with the UVI system in the Virgin Islands and in Alberta, Canada.
The UVI system is capable of producing approximately 11,000 pounds of tilapia and 1,400 cases of lettuce or 11,000 pounds of basil annually based on studies in the Virgin Islands. Enterprise budgets for tilapia production combined with either lettuce or basil have been developed. The U.S. Virgin Islands represent a small niche market with very high prices for fresh tilapia, lettuce and basil, as more than 95 percent of vegetable supplies and nearly 80 percent of fish supplies are imported. The budgets were prepared to show revenues, costs and profits from six production units. A commercial enterprise consisting of six production units is recommended because one fish-rearing tank (out of 24) could be harvested weekly, thereby providing a continuous supply of fish for market development.
The enterprise budget for tilapia and lettuce shows that the annual return to risk and management (profit) for six production units is $185,248. The sale prices for fish ($2.50 per pound) and lettuce ($20.00 per case) have been established through many years of market research at UVI. Most of the lettuce consumed in the Virgin Islands is imported from California. It is transported by truck across the United States to East Coast ports and then shipped by ocean freighters to Caribbean islands. Local production capitalizes on the high price of imports caused by transportation costs. Locally produced lettuce is also fresher than imported lettuce. Although this enterprise budget is unique to the U.S. Virgin Islands, it indicates that aquaponic systems can be profitable in certain niche markets.
The enterprise budget for tilapia and basil shows that the annual return to risk and management for six production units is $693,726. Aquaponic systems are very efficient in producing culinary herbs such as basil (Figure 11) and a conservative sale price for fresh basil with stems in the U.S. Virgin Islands is $10.00 per pound. However, this enterprise budget is not realistic in terms of market demand. The population (108,000 people) of the U.S. Virgin Islands cannot absorb 66,000 pounds of fresh basil annually, although there are opportunities for provisioning ships and exporting to neighboring islands. A more realistic approach for a sixunit operation is to devote a portion of the growing area to basil to meet local demand while growing other crops in the remainder of the system.
Figure 11. Basil production in the UVI aquaponic system.
The break-even price for the aquaponic production of tilapia in the Virgin Islands is $1.47 per pound, compared to a sale price of $2.50 per pound. The break-even prices are $6.15 per case for lettuce (sale price = $20.00 per case) and $0.75 per pound for basil (sale price = $10.00 per pound). The break-even prices for tilapia and lettuce do not compare favorably to commodity prices. However, the cost of construction materials, electricity, water, labor and land are very high in the U.S. Virgin Islands. Break-even prices for tilapia and lettuce could be considerably lower in other locations. The break-even price for basil compares favorably to commodity prices because fresh basil has a short shelf life and cannot be shipped great distances.
A UVI aquaponic system in an environmentally controlled greenhouse at the Crops Diversification Center South in Alberta, Canada, was evaluated for the production of tilapia and a number of plant crops. The crops were cultured for one production cycle and their yields were extrapolated to annual production levels. Based on prices at the Calgary wholesale market, annual gross revenue was determined for each crop per unit area and per system with a plant growing area of 2,690 square feet (Table 2).
Table 2. Preliminary production and economic data from the UVI aquaponic system at the Crop Diversification Center South, Alberta, Canada.1 (Data courtesy of Dr. Nick Savidov).
| Crop | Annual production lb/ft2 | Annual production tons/2,690 ft2 |
Wholesale price Unit |
|---|---|---|---|
| Tomatoes | 6. | 8.1 | 15 lb |
| Cucumbers | 12.4 | 16.7 | 2.2 lb |
| Eggplant | 2.3 | 3.1 | 11 lb |
| Genovese basil | 6.2 | 8.2 | 3 oz |
| Lemon basil | 2.7 | 3.6 | 3 oz |
| Osmin basil | 1.4 | 1.9 | 3 oz |
| Cilantro | 3.8 | 5.1 | 3 oz |
| Parsley | 4.7 | 6.3 | 3 oz |
| Portulaca | 3.5 | 4.7 | 3 oz |
| Crop | Wholesale price $ |
Total value $/ft2 |
Total value $/2,690 ft2 |
|---|---|---|---|
| Tomatoes | 17.28 | 6.9 | 18542 |
| Cucumbers | 1.58 | 8.9 | 23946 |
| Eggplant | 25.78 | 5.33 | 14362 |
| Genovese basil | 5.59 | 186.64 | 502044 |
| Lemon basil | 6.31 | 90.79 | 244222 |
| Osmin basil | 7.03 | 53.23 | 143208 |
| Cilantro | 7.74 | 158.35 | 425959 |
| Parsley | 8.46 | 213.81 | 575162 |
| Portulaca | 9.17 | 174.2 | 468618 |
1 Economic data based on Calgary wholesale market prices for the week ending July 4, 2003.
Annual production levels based on extrapolated data from short production cycles are subject to variation. Similarly, supply and demand will cause wholesale prices to fluctuate during the year. Nevertheless, the data indicate that culinary herbs in general can produce a gross income more than 20 times greater than that of fruiting crops such as tomatoes and cucumbers. It appears that just one production unit could provide a livelihood for a small producer. However, these data do not show capital, operating and marketing costs, which will be considerable. Furthermore, the quantity of herbs produced could flood the market and depress prices. Competition from current market suppliers will also lead to price reductions.
Overview
Although the design of aquaponic systems and the choice of hydroponic components and fish and plant combinations may seem challenging, aquaponic systems are quite simple to operate when fish are stocked at a rate that provides a good feeding rate ratio for plant production. Aquaponic systems are easier to operate than hydroponic systems or recirculating fish production systems because they require less monitoring and usually have a wider safety margin for ensuring good water quality. Operating small aquaponic systems can be an excellent hobby. Systems can be as small as an aquarium with a tray of plants covering the top. Large commercial operations comprised of many production units and occupying several acres are certainly possible if markets can absorb the output. The educational potential of aquaponic systems is already being realized in hundreds of schools where students learn a wide range of subjects by constructing and operating aquaponic systems. Regardless of scale or purpose, the culture of fish and plants through aquaponics is a gratifying endeavor that yields useful products—food.
The information given herein is for educational purposes only. Reference to commercial products or trade names is made with the understanding that no discrimination is intended and no endorsement by the Southern Regional Aquaculture Center or the Cooperative Extension Service is implied.
SRAC fact sheets are reviewed annually by the Publications, Videos and Computer Software Steering Committee. Fact sheets are revised as new knowledge becomes available. Fact sheets that have not been revised are considered to reflect the current state of knowledge.
The work reported in this publication was supported in part by the Southern Regional Aquaculture Center through Grant No. 2003-38500-12997 from the United States Department of Agriculture, Cooperative State Research, Education, and Extension Service.
James E. Rakocy
Agricultural Experiment Station
University of the Virgin Islands
Michael P. Masser
Department of Wildlife and Fisheries Sciences
Texas A&M University
Thomas M. Losordo
Biological and Agricultural Engineering Department
North Carolina State University
Marley Beem
Extension Aquaculture Specialist