Fungicide Resistance Management
Fungicides are important tools for managing diseases in many crops. Unlike insecticides and some herbicides which kill established insects or weeds, fungicides are most commonly applied to protect healthy plants from infection by fungal plant pathogens. To be effective, fungicides must be applied before infections become established and in a sufficient spray volume to achieve thorough coverage of the plant or treated area. Protection from fungicides is temporary because they are subject to weathering and breakdown over time. They also must be reapplied to protect new growth when disease threatens. Poor disease control with fungicides can result from several causes including insufficient application rate, inherently low effectiveness of the fungicide on the target pathogen, improper timing or application method, and excessive rainfall. Resistance (lack of sensitivity) to fungicides in fungal pathogens is another cause of poor disease control. The development of fungicide resistance is influenced by complex interactions of factors such as the mode of action of the fungicide (how the active ingredient inhibits the fungus), the biology of the pathogen, fungicide use pattern, and the cropping system. Understanding the biology of fungicide resistance, how it develops, and how it can be managed is crucial for insuring sustainable disease control with fungicides.
The problem of fungicide resistance became apparent following the registration and widespread use of the systemic fungicide (see fungicide mobility below) benomyl (Benlate) in the early 1970’s. Prior to the registration of benomyl, growers routinely applied a protectant fungicide (see fungicide mobility below) such as maneb, mancozeb, or copper to control diseases without experiencing resistance problems. A distinct advantage of benomyl over the protectant fungicides was its systemic activity. In addition to protecting plants from infection, systemic activity conferred rainfastness and provided disease control when applied after the early stages of infection. Superior disease control was often achieved with benomyl compared to the protective dithiocarbamates. However, benomyl differed from the dithiocarbamates in its site-specific mode of action (see Fungicide Groups and Mode of Action below) which was readily overcome by several fungal pathogens. Resistance problems appeared a few years after benomyl was introduced where the fungicide was used intensively. Sudden control failures occurred with diseases such as powdery mildew, peanut leaf spot, and apple scab.
Many of the fungicides developed and registered since the introduction of benomyl also are systemic, have a site-specific mode of action, and are at increased risk for resistance problems (Table 1). Fungicide resistance is now a widespread problem in global agriculture. Fungicide resistance problems in the field have been documented for nearly 200 diseases (crop – pathogen combinations), and within about half of the known fungicide groups. Many more cases of resistance are suspected but have not been documented. While resistance risks with many of fungicides may not be as great as with benomyl, strategies to manage the resistance risk have been developed and implemented to avoid unexpected control failures and sustain the usefulness of new products. As a result of resistance management strategies, fungicides within all mode of action groups remain useful disease management tools in at least some cropping systems. The purpose of this bulletin is to describe the resistance phenomenon, identify resistance risks in the different fungicide groups, and to provide general guidelines for managing resistance. Since this fact sheet was first written, many new fungicides have been registered, and mode of action groups and specific resistance management strategies are now specified on fungicide labels. The listing of fungicides by mode of action group here (Table 1) is useful for identifying appropriate fungicides for use in tank mixtures and application schedules as part of the recommended resistance management programs.
Fungicide Mobility
Understanding the mobility of fungicides on and in treated plants, and how various fungicides are classified based on mobility is important when making decisions pertaining to the selection of the best of fungicide for a particular disease and its optimal application timing. Fungicides can be classified into two basic mobility groups: protectant or penetrant. Regardless of its mobility characteristics, no fungicide will be highly effective after the development of disease symptoms and pathogen reproduction (spore production). Fungicides can slow or stop the development of new symptoms if applied in a timely fashion, but fungicides will not cure existing disease symptoms. Therefore, understanding fungicide mobility, fungicide mode of action, and the biology of the target pathogen are important so that fungicide applications are made before the disease becomes established and more difficult to control.
Protectant fungicides are active on the plant surfaces where they remain after application. There is no movement of the fungicide into the plant. Because they remain on the plant surface, protectant fungicides loose activity after being washed off the plant and must be re-applied to new growth that develops after application. Protectant fungicides typically prevent spore germination, therefore they must be applied prior to infection and have no effect once the fungus grows into the plant resulting in infection.
Penetrant fungicides are absorbed into plants following application. Because these fungicides are absorbed into plants, they are generally considered systemic fungicides. However, penetrant fungicides have different degrees of systemic movement once inside the plant. Some fungicides are ‘locally systemic’, only moving a short distance such as through a few layers of plant cells. Fungicides that move from one side of a leaf to other have ‘translaminar’ movement. Translaminar and locally systemic fungicides are not transported throughout the plant. Highly mobile fungicides are either ‘xylem-mobile’ or ‘true systemics’. Xylem-mobile fungicides move upward in plants and outward to the periphery of leaves with water through the xylem, the water conducting tissue of the plant. True systemic fungicides move both upward through the xylem, and downward through the phloem, the food conducting tissue of the plant. Few if any fungicides are fully systemic. Unlike protectant fungicides, penetrant fungicides are rain fast within a few hours of application and may require less thorough application coverage to be effective. In addition, many penetrant fungicides inhibit fungal growth and sporulation and can be effective when applied after the early stages of infection.
Regardless of the level of systemic movement, penetrant fungicides have limited ‘curative’ ability. Generally they only stop or slow infections within the first 24 to 72 hour period following fungal penetration into the plant. Therefore, penetrant fungicides must be applied before or shortly after infection, and are ineffective on existing symptoms. Both protectant and penetrant fungicides provide good disease control when applied before infection and are best applied on a preventive schedule.
Development of Fungicide Resistance
Resistance is a genetic adjustment by a fungus that results in reduced sensitivity to a fungicide. Reduced sensitivity is thought to be a result of genetic mutations which occur at low frequencies (one in a million or less) or of naturally occurring sub-populations of resistant individuals. Individuals in a fungal population may consist of the mycelium (the body of a fungus), sclerotia (large survival structures), spores (small reproductive structures), or the nucleus of single cells capable of reproduction and spread. The resistance trait may result from single gene or multiple gene mutations (see build-up of resistance below). Single-gene mutations that confer resistance to site-specific fungicides are more likely to develop than the simultaneous occurrence of mutations in multiple genes needed to confer resistance to multi-site inhibiting fungicides. Mechanisms of resistance differ depending on the mode of action, but include alteration of the target site, reduced fungicide uptake, active export of the fungicide outside fungal cells, and detoxification or breakdown of the fungicide.
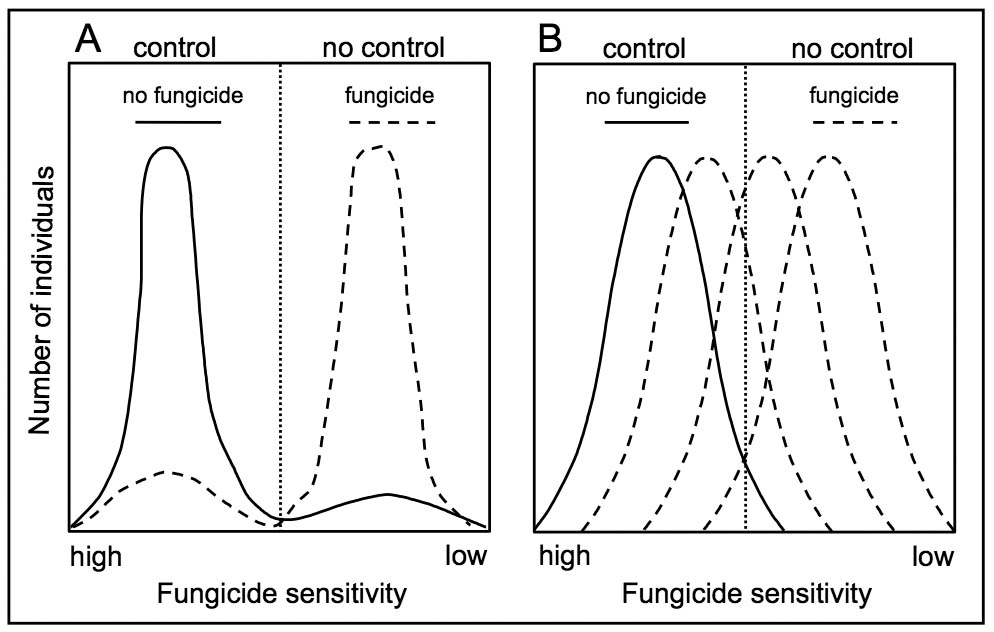
The level of resistance to a fungicide can be measured in the laboratory by exposing a collection of members of a field population to the fungicide and measuring toxicity response. Toxicity responses are usually measured as inhibition of fungus growth, spore germination, or actual plant infection in cases where the fungus cannot be cultured. The effective concentration which inhibits growth, germination, or infection by 50% (EC50) is then calculated for each sampled individual much in the same way an LD50 (50% lethal dose) is calculated for assessing the acute toxicity of a pesticide to rats or mice. Where many members of a population are sampled and screened, a range of sensitivity (or resistance) to the fungicide is usually observed. The frequency distribution of the sensitivity of individuals in the population is usually normal or bell-shaped, typical of many biological responses in nature (Figure 1). Where the fungicide is newly introduced or where the risk of resistance is low, the population is distributed over a sensitive range. However, a distribution consisting of two distinct sub-populations also may occur where a small sub-population of resistant strains is present along with a larger sub-population of sensitive strains (Figure 1A).
Build-up of Resistance
Resistance in a population becomes important when the frequency of resistant strains builds up to dominate the population. The build-up of resistant strains is caused by repeated use of the fungicide which exerts selection pressure on the population. The fungicide selectively inhibits sensitive strains, but allows the increase of resistant strains. This shift toward resistance occurs at different rates depending on the number of genes conferring resistance. When single gene mutations confer resistance, a rapid shift toward resistance may occur, leading to a population that is predominantly resistant and where control is abruptly lost (Figure 1A). When multiple genes are involved, the shift toward resistance progresses slowly, leading to a reduced sensitivity of the entire population (Figure 1B). The gradual shift with the multiple gene effect may result in reduced fungicide activity between sprays, but the risk of sudden and complete loss of control is low. It is difficult to clearly distinguish between sensitive and resistant sub-populations with field sampling during the early shifts towards reduced sensitivity because sensitivity responses overlap. Large numbers of individuals must be tested to identify the gradual type of resistance.
Assessing Resistance Risk
Many factors affect the development of resistance and its build-up in the field, which makes it difficult to predict the resistance risk for new fungicides. Despite resistance problems that have been identified following the introduction of some new fungicides, many examples can be cited where their use continues to be effective. Factors that must all be considered in assessing resistance risk include the properties of the fungicide, the biology of the pathogen, and the crop production system where the fungicide is used.
Fungicide Groups and Mode of Action
Fungicides are grouped by similarities in chemical structure and mode of action (Table 1). Site-specific fungicides disrupt single metabolic processes or structural sites of the target fungus. These include cell division, sterol synthesis, or nucleic acid (DNA and or RNA) synthesis. The activity of site-specific fungicides may be reduced by single or multiple-gene mutations. The MBC (benzimidazole), PA (phenylamide), and QoI (strobilurin) groups are subject to single-gene resistance and carry a high risk of resistance problems. Other groups with site-specific modes of action include the Dicarboximides and DMIs (sterol demethylation inhibitors), but resistance to these fungicides appears to involve slower shifts toward insensitivity because of multiple-gene involvement. Many of the site-specific fungicides also have systemic mobility. However, systemic mobility is not necessary for resistance development. Resistance problems have developed in the dicarboximide group and with dodine which are protectant fungicides.
Multi-site fungicides interfere with many metabolic processes of the fungus and are usually protectant fungicides. Once taken up by fungal cells, multisite inhibitors act on processes such as general enzyme activity that disrupt numerous cell functions. Numerous mutations affecting many sites in the fungus would be necessary for resistance to develop. Typically these fungicides inhibit spore germination and must be applied before infection occurs. Multi-site fungicides form a chemical barrier between the plant and fungus. The risk of resistance to these fungicides is low.
There are two codes currently used to classify fungicides by mode of action (Table 1). The mode of action group (A, B, etc.) refers to the general target site in the pathogen such as nucleic acid synthesis, cell wall synthesis, respiration, etc. Sub-groups (A1, A2, etc.) within a mode of action group refer to specific biochemical target sites of fungicide activity. The FRAC (Fungicide Resistance Action Committee) code is used on most fungicide labels. The FRAC code refers to fungicides that have same site-specific mode of action and share the same resistance problems across members of the group (cross-resistance). FRAC groups are currently numbered from 1 to 46 in order of their introduction to the marketplace. FRAC groups and mode of action subgroups are mostly the same.
Table 1. Fungicides registered in the United States grouped by mode of action and relative risk for developing resistance problems.
| Mode of action | Group1 | Group name | Common name | Trade names2 | Mobility3 | Uses4 | Risk5 |
|---|---|---|---|---|---|---|---|
| Nucleic acid synthesis | A1 (4) | PA | metalaxy | Allegiance, MetaStar, | S | ST, F, S | H |
| mefenoxam or metalaxyl-M | Ridomil Gold, Apron XL, Subdue, Ultra Flourish | S | ST, F, S | H | |||
| Mitosis and cell division | B1 (1) | MBC | thiabendazole | Mertect | S | ST, PH | H |
| thiophanate-methyl | Topsin M, Cleary’s 3336, OHP 6672 | S | ST, F, S | H | |||
| B3 (22) | Benzamides | zoxamide | Gavel (pre-mix) | S | F | M | |
| B5 (43) | Benzamide | fluopicolide | Presidio, Adorn | S | F, S | M | |
| Respiration | C2 (7) | SDHI | boscalid | Endura, Emerald | S | F, S | M-H |
| carboxin | Vitavax | S | ST | L | |||
| flutolanil | Moncut, ProStar | S | ST, F, S | M | |||
| fluopyram | Propulse, Luna (pre-mixes) | S | F, S | M-H | |||
| fluxapyroxad | Acceleron DX612, Priaxor, Merivin (pre-mixes) | S | ST,F,S | M-H | |||
| penflufen | EverGol (pre-mixes) | S | ST, S | L | |||
| penthiopyrad | Fontelis, Velista, Vertisan | S | F, S | M-H | |||
| sedexane | Vibrance | S | ST | L | |||
| C3 (11) | Strobilurin (QoI) | azoxystrobin | Abound, Heritage, Quadris, Dynasty | S | F, S, ST | H | |
| famoxidone | Tanos (pre-mix) | S | F | H | |||
| fenamidone | Reason, Fenstop, Idol | S | F, ST | H | |||
| fluoxastrobin | Aftershock, Evito, Disarm | S | F, S | H | |||
| kresoxim-methyl | Cygnus, Sovran | S | F | H | |||
| picoxystrobin | Approach | S | F | H | |||
| pyraclostrobin | Cabrio, Insignia, Headline, Acceleron DX109 | S | F, S, ST | H | |||
| trifloxystrobin | Flint, Compass, Gem, Trilex | S | F, S, ST | H | |||
| C4 (21) | QiI | cyazofamid | Ranman, Segway | S | F | M | |
| C5 (29) | Dinitroaniline | fluazinam | Omega, Secure | P | F, S | L | |
| C6 (30) | Organo tin | triphenyl tin hydroxide | Super Tin, Agri Tin | P | F | L | |
| C8 (45) | QxI | ametoctradin | Orvego, Zampro (pre-mixes) | P | F, S | M-H | |
| Amino acids and proteins | D1 (9) | AP | cyprodinil | Vanguard; Switch, Inspire, Super, Palladium (pre-mixes) | S | F | M |
| pyrimethanil | Scala | S | F | M | |||
| D4 (25) | Antibiotic (bactericide) | streptomycin | Agri-Mycin, Firewall | P | ST, F | H | |
| D5 (41) | Antibiotic (bactericide) | oxytetracycline | Mycoshield, Fireline | P | F | H | |
| Signaling | E1 (13) | Aza-naphthalenes | quinoxyfen | Quintec | P | F | M |
| E2 (12) | PP | fludioxonil | Maxim, Scholar, Medallion | P | ST, F, PH | L-M | |
| E3 (2) | Dicarboximide | iprodione | Rovral, Chipco 26019, 26GT | P | F, S | M-H | |
| vinclozolin | Curalan | P | F, S | M-H | |||
| Lipids and membranes | F3 (14) | Aromatic Hydrocarbon | dichloran | Botran | P | F, S, PH, ST | L-M |
| PCNB | Terraclor, Turfcide, Blocker | P | ST, S | L | |||
| etridiazole | Terrazole, Terramaster, Truban | P | S | L-M | |||
| F4 (28) | Carbamate | propamocarb HCl | Previcur Flex, Banol | S | F, S | L-M | |
| Sterol synthesis | G1 (3) | DMI (SBI: Class I) | cyproconazole | Alto | S | F | M |
| fenarimol | Rubigan | S | F, S | M | |||
| imazalil | Raxil,Vibrance (pre-mixes) | S | ST | L | |||
| difenoconazole | Inspire Super MP, Top MP, Dividend and other pre-mixes | S | ST, F | L-M | |||
| fenbuconazole | Enable, Indar | S | F | M | |||
| ipconazole | Rancona, various pre-mixes | S | ST | L | |||
| myclobutanil | Rally, Eagle, Laredo, Spera | S | ST, F, S | L-M | |||
| propiconazole | Tilt, Orbit, Banner Maxx, various generic brands and pre-mixes | S | F, S | M | |||
| prothioconazole | Proline; Prosaro, Provost, and EverGol premixes | S | ST, F, S | L-M | |||
| tebuconazole | Folicur, Elite, Raxil numerous generics and pre-mixes | S | F, S, ST | L-M | |||
| tetraconazole | Domark, Eminent | S | F | M | |||
| triadimefon | Bayleton | S | F, S | M | |||
| triadimenol | Trilex (pre-mix) | S | ST | L | |||
| triflumizole | Procure, Terraguard | S | F, S | M | |||
| triticonazole | Charter, Trinity, Triton | S | ST, F, S | L-M | |||
| G3 (17) | SBI: Class III | fenhexamid | Elevate, Decree | P | F | L-M | |
| Cell wall synthesis | H4 (19) | Polyoxin | polyoxin | Affirm, Endorse, Ph-D, Veranda | S | F, S | M |
| H5 (40) | CAA | dimethomorph | Forum, Stature, premixes | S | F, S | L-M | |
| mandipropamid | Revus, Micora, Revus Top (pre-mix) | S | F, S | L-M | |||
| Plant defense activator | P1 (P1) | BTH | acibenzolar-S-methyl | Actigard | S | F | L |
| Unknown | U1 (27) | Cyanoacetamideoxime | cymoxanil | Curzate, Tanos (pre-mix) | S | F | M |
| U2 (33) | Phosphonate | fosetyl-AL | Aliette, Signature | S | F | L | |
| phosphorous acid | Agri-Fos, Alude, Appear, Fosphite, KPhyte, Phostrol, | ||||||
| Prophyt, Rampart, Vital | S | F | L | ||||
| U8 (U8) | Aryl-phenyl-ketone | metrafenone | Vivando | S | F | M | |
| U12 (U12) | Guanadine | dodine | Syllit | P | F | M | |
| Multi-site activity | M1 (M1) | Inorganic | copper salts | Kocide, Cuprofix, Champ, Nu-Cop, Cuproxat | P | F | L |
| M2 (M2) | Inorganic | sulfur | Microthiol, Sulfur | P | F | L | |
| M3 (M3) | Dithiocarbamate | ferbam | Ferbam | P | F | L | |
| mancozeb | Dithane, Penncozeb, Manzate, Fore | P | F, ST | L | |||
| metiram | Cabrio plus (pre-mix) | P | F | L | |||
| thiram | Thiram, Defiant, Signet, Spotrete | P | F, ST | L | |||
| ziram | Ziram | P | F | L | |||
| M4 (M4) | Phthalimide | captan | Captan, Captec | P | F, ST | L | |
| M5 (M5) | Chloronitrile | chlorothalonil | Bravo, Daconil, generics and premixesnumerous | P | F, S | L |
Fitness of Resistant Strains
Fitness is the ability to compete and survive in nature. Strains of pathogens resistant to some fungicides compete equally well with sensitive strains and are still present after the fungicide in question is no longer in use. For example, strains of Cercospora arachidicola which cause early leaf spot of peanut are still established in the southeastern U.S. where benomyl resistance was a problem over 20 years ago. Therefore, fungicides with resistance problems cannot be successfully reintroduced into areas where resistant strains are highly fit. Fortunately, resistant strains are sometimes less fit than wild-type sensitive strains. This has been true for DMI resistance in powdery mildews and for dicarboximide resistance in Botrytis diseases. Unfit strains only compete well under the selection pressure of the fungicide. Thus, the resistance is at least partially reversible when the selection pressure of the fungicide is removed or minimized by using resistance management.
Fungicide Use Pattern
Frequent and exclusive usage of at-risk fungicides increases the risk of resistance problems. Selection pressure is increased where repeated applications are required for disease control as with many foliar diseases. Selection pressure and the risk of resistance are low for seed treatments and for many soilborne diseases which require only one or two applications per season. The method and rate of application may also impact resistance development. Poor disease control resulting from causes such as improper application timing or inadequate spray coverage may result in a need for a more intensive spray program and the exposure of more individuals to the fungicide. Using adequate rates in a manner that produces good disease control reduces the reproductive capacity of fungal pathogens, thus reducing selection pressure. Similarly, a preventive spray program is less risky than a rescue program because selection pressure is applied to fewer individuals. Finally, an increase in selection pressure results from an excessive number of applications where a real need is not justified.
Pathogen Biology
Fungal pathogens with high rates of reproduction are most prone to develop fungicide resistance. Because many individuals (usually spores) are produced by these fungi, more individuals are exposed to selection pressure and there is a greater probability of mutations that lead to reduced fungicide sensitivity. Foliar diseases produce thousands of spores on the surface of an individual leaf spot. Furthermore, these diseases typically have several reproductive cycles per season. Under selection pressure of a fungicide, resistant individuals may increase rapidly and dominate the population after several cycles of infection and reproduction.
Diseases with low reproduction rates generally complete only one life cycle per season. Soilborne pathogens produce fewer offspring per season than their foliar counterparts. Some soilborne diseases reproduce by forming seed-like survival structures called sclerotia. There may be fewer than a hundred sclerotia formed per plant. Where an at-risk fungicide is used for soilborne disease control, resistance development is likely to be slow because comparatively few individuals are exposed to selection pressure.
Crop Production Practices
Production practices that favor increased disease pressure also promote resistance development by increasing the number of individuals exposed to selection pressure. Pathogens reproduce at higher rates on susceptible varieties compared to resistant or partially resistant varieties. Selection pressure also may be reduced for resistant varieties because fewer applications should be needed for effective disease control. Inadequate or excessive fertilization with nitrogen may increase disease incidence in some crops. For example, early blight of potato and tomato and dollar spot of turfgrass are favored by nitrogen deficiency. Alternatively, the severity of spring dead spot of bermudagrass and some foliar diseases of wheat is increased with intensive nitrogen fertilization. Excessive irrigation or frequent irrigation with small amounts of water increases the incidence of many diseases by promoting disease spread, extended periods of leaf wetness, and high soil moisture.
Continuous cropping and poor sanitation practices promote severe early-season disease development. Closed cropping systems such as greenhouses are particularly prone to resistance problems because plants are grown in crowded conditions that may favor severe disease development, rapid spread, and high selection pressure. Permanently established plantings of perennial crops such as orchards, nurseries, and vineyards are also prone to resistance problems. Unlike annual crops where crop rotation can be practiced, many pathogens survive from year to year on plants and crop debris within permanent plantings resulting in a local pathogen population exposed to yearly selection pressures.
Resistance Management Strategies
Strategies for managing fungicide resistance are aimed at delaying its development. Therefore, a management strategy should be implemented before resistance becomes a problem. The only way to absolutely prevent resistance is to not use an at-risk fungicide. This is not a practical solution because many of the modern fungicides that are at risk for resistance problems provide highly effective, broad-spectrum disease control. By delaying resistance and keeping its level under control, resistance can be prevented from becoming economically important. Because practical research in the area of fungicide resistance management has been limited, many of the strategies devised are based in the theory of expected responses of a pathogen population to selection pressure. For the most part, evaluations of the effectiveness of these strategies have not been based on research, but rather on observations made where the fungicides have been used commercially on a large scale.
Specific strategies for resistance management vary for the different fungicide groups,
the target pathogen(s), and the crop. However, some strategies are generally effective.
Resistance management should integrate cultural practices and optimum fungicide use
patterns. The desired result is to minimize selection pressure through a reduction
in time of exposure or the size of the population exposed to the at-risk fungicide.
Probably the most important aspect of optimizing use patterns is the deployment of
tank mixtures and alternating sprays of the at-risk fungicide with a fungicide from
a different mode of action group. The comparative merits of tank-mixing compared
to alternating sprays have been debated. Some theorize that tank-mixing reduces selection
pressure only when the partner fungicide is highly effective and good coverage is
achieved. Alternating fungicides is thought to act by reducing the time of exposure.
In practice, examples can be cited for the effectiveness of both approaches. Both
practices are more effective when cultural practices are implemented to reduce disease
pressure. The alternation of blocks of more than one spray is probably less effective
in resistance management than the other use patterns. For example, a block of four
continuous sprays of the DMI fungicide tebuconazole is recommended at mid-season for
peanut disease control. Despite the use of at least one application of a non-DMI
fungicide before and after the 4-spray block, resistance to tebuconazole in both early
and late leaf spot diseases became a widespread problem in less than 10 years.
The proper choice of a partner fungicide in a resistance management program is critical. Generally, good partner fungicides are multi-site inhibitors that have a low resistance risk (e.g. chlorothalonil, mancozeb, etc.) and are highly effective against the target pathogen. However, the use of an unrelated at-risk fungicide with no potential for cross-resistance problems also may be effective. Numerous fungicides are now marketed as pre-mixtures of two or more fungicide groups. These are convenient for resistance management, but users should be mindful of the fungicide groups in the pre-mix, so an at-risk group is not over-extended. Characteristics of important fungicide groups and their resistance risks are discussed below.
MBCs (FRAC Group 1; Mode of Action Sub-Group B1)
MBC (Methyl Benzimidazole Carbamate) fungicides are site-specific fungicides which interfere with cell division. They have systemic mobility and have activity on many pathogens except water molds (e.g. Pythium and Phytophthora) and darkly pigmented fungi (e.g. Alternaria). Research has demonstrated that benzimidazole resistant strains may be present at low frequencies in nature, even in the absence of fungicide exposure. Under selection pressure, resistance development is abrupt and rapid (Figure 1A). Resistant strains cannot be controlled by increasing the application rate or by shortening the spray interval. Resistant strains are often fit and competitive in nature even without selection pressure. Therefore, some populations have remained resistant where benzimidazole use has been discontinued for 10 years. Resistance to benzimidazoles has been documented for over 70 diseases and cross-resistance exists within this fungicide group. Benzimidazole resistance has received less recent attention because the fungicide benomyl is no longer registered in the U.S. However, resistance management remains important for thiophanate-methyl, the other widely used benzimidazole fungicide. In spite of the numerous resistance problems with benzimidazoles, there are also many examples where benzimidazoles have remained effective for over 30 years with judicial use.
Strobilurins (FRAC Group 11; Mode of Action Sub-Group C3)
Strobilurin fungicides, also know as quinone-outside inhibitor (QoI) fungicides, are synthetic analogues of a naturally occurring compound produced by a wood rotting fungus. Strobilurins inhibit respiration in fungal cells by targeting a protein (cytochrome bc-1) that is encoded by a gene in the mitochondria. The fungicides are broad-spectrum with activity against all the major types of fungal pathogens. Strobillurin fungicides penetrate plant leaves and move from one side of the leaf to the other. This translaminar mobility makes them rain-fast, but they lack true systemic movement in the plant compared to some other fungicide groups. Strobilurins act on a broad range of fungal processes including spore germination, fungal growth, and reproduction (sporulation). Strobilurin fungicides have been registered on numerous crops because of their broad-spectrum activity and excellent human and environmental safety profiles. However, like the benzmidazoles, resistance developed shortly after their introduction in the late 1990’s. Three different single-gene mutations have been identified that abruptly confer resistance (Figure 1A) that has been documented for more than 50 diseases. Resistance has been documented for diseases such as Septoria leaf spot on wheat; gummy stem blight, downy mildew, and powdery mildew on cucurbits, and frogeye leaf spot on soybeans. Resistant isolates are cross-resistant to all other strobilurin fungicides, but not to other mode of action groups including the closely related QiI (Group C4 or 21) fungicides. Several strobilurin fungicides are marketed in pre-mixtures with non-strobilurin fungicides for use on certain crops.
Dicarboximides (FRAC Group 2; Mode of Action Sub-Group E3)
Dicarboximides inhibit both spore germination and fungal growth. Resistance is thought to arise by mutations. The frequency of resistant individuals and their level of resistance increase gradually with prolonged selection pressure (Figure 1B). Resistance to dicarboximide fungicides has been identified for more than 15 diseases including brown rot of stone fruits, gray mold (Botrytis) on several crops, and important turf grass diseases. Dicarboximide resistant strains of some pathogens are less fit to survive than sensitive strains. Reduced exposure of resistant strains to dicarboximide fungicides results in a decrease in the frequency of resistant strains and possibly an overall shift of the population back toward sensitivity. Thus, it has been possible to reintroduce dicarboximides into problem situations where resistance management has been implemented.
DMIs (FRAC Group 3; Mode of Action Sub-Group G1)
Demethylation inhibitor (DMI) fungicides are site-specific fungicides that disrupt the synthesis of sterols. Sterols are compounds required for growth of many plant pathogenic fungi. DMIs are a large group of systemic fungicides that have a broad range of activity against many types of foliar and soilborne diseases except for those caused by the water molds. Typically, resistance develops gradually as a result of an accumulation of several mutations (polygenic resistance) and is at first difficult to detect (Figure 1B). Resistant strains are thought to have reduced fitness; therefore, reduced selection pressure through the use resistance management strategies may partially shift the resistant populations back toward sensitivity. DMI resistance has been documented for over 30 diseases including apple scab, powdery mildews, gray mold, and brown rot of stone fruit.
Pre-mixtures of DMI fungicides with strobilurin or protectant fungicides are being marketed for many crops to improve the spectrum of diseases controlled and to comply with resistance management guidelines.
PAs (FRAC Group 4; Mode of Action Sub-Group A1)
PA (Phenylamide) fungicides are water soluble, highly systemic fungicides specifically used to control diseases caused by water molds. Such diseases include damping off and root and lower stem rots caused Pythium and Phytophthora, and foliar diseases such as late blight, downy mildew, and white rust. PA fungicides inhibit fungal growth by disrupting RNA synthesis. Resistance problems with PAs, specifically metalaxyl, were observed shortly after their introduction where they were used exclusively and disease pressure was high. Resistance, now confirmed for over 30 diseases, is governed by one or two genes and a low frequency of resistant individuals may exist in wild populations prior to use of these fungicides. Resistance can increase rapidly through selection of the naturally occurring strains (Figure 1A). Cross resistance occurs with other PA fungicides, but not with fungicides from other mode of action groups. Both resistant and sensitive strains survive in the absence of PA fungicide use and their levels tend to equilibrate over time. Resistance management is critical to limit the proportion of resistant strains in a population. The manufacturer of metalaxyl-M markets premixes with mancozeb, copper, and chlorothalonil for use against foliar pathogens. The marketing of pre-mixes of metalaxyl-M with non-related protectant fungicides ensures compliance with a resistance management strategy.
SDHIs (FRAC Group 7; Mode of Action Sub-Group C2)
SDHI (Succinate Dehydrogenase Inhibitor) fungicides inhibit respiration in fungal pathogens by blocking an important enzyme in mitochondrial respiration. SDHI fungicides are systemic and control a broad spectrum of disease. While the fungicide carboxin has been registered for over 40 years, several newer generation SDHI fungicides have been recently registered for use on a wide range of crops. SDHI fungicides are used as seed treatments as well as foliar applied treatments. They have activity on a range of foliar diseases (rusts, powdery mildews, leaf spots) and soilborne diseases cause by Sclerotinia and Rhizoctonia. Resistance development, caused by several point mutations in the succinate dehydrogenase gene, has been documented for over 10 diseases such as gummy stem blight and powdery mildew on cucurbits, and grey mold on grapes and strawberries. Despite the few cases of documented resistance, the group has a moderate to high resistance risk and resistance management should be implemented for disease management programs that require multiple applications.
Conclusions
Fungicide resistance is one of several possible causes of poor disease control. Fungicide resistance not only threatens the usefulness of individual fungicides, but also the farm economy because of potential yield losses from poor disease control. Unfortunately, registrations are being lost for older broad-spectrum fungicides that have a low resistance risk. Many of the newer replacement fungicides are more selective in the number and types of diseases controlled and have site-specific modes of action making them more prone to resistance problems. Maintaining an array of effective fungicides is critical. Resistance management strategies should be recommended by crop advisors and implemented by growers to prolong the active life of at-risk fungicides. Fungicide groups have different levels of resistance risk. Risk assessment is critical for newly developed fungicides. Mode of action group and resistance management strategies are now clearly included on the registration labels of most site-specific fungicides. However, it is difficult to predict the actual risk of resistance because of many interacting factors. Experience with fungicide resistance indicates that resistance problems are often manageable. Monitoring resistance levels in pathogen populations is essential for assessing risk and evaluating management practices. Unfortunately, there is no coordinated monitoring effort in place and growers will generally have to rely on proven methods of resistance management.
References
1) Beckerman, J. 2008. Understanding fungicide mobility. Purdue Extension BP-70-W.
2) Lyr, H. 1995. Modern selective fungicides: properties, applications, mechanisms
of action. Jena, New York; Gustav Fischer, Deerfield Beach, Fla.; 595 p.
3) Fungicide Resistance Action Committee (http://www.frac.info).
John Damicone
Extension Plant Pathologist
