Arthropod Pest Management in Greenhouses and Interiorscapes
Insects and their relatives cause major plant damage in commercial greenhouses and interiorscapes. Identification of key pests and an understanding of appropriate control measures are essential to guard against costly crop losses. With tightening regulations on conventional insecticides and increasing consumer sensitivity to their use in public spaces, growers must seek effective pest management alternatives to conventional chemical control. Management strategies centered around the concept of integrated pest management (IPM) can provide growers powerful tools for preventing pest problems before they occur or at least mitigate their ornamental/economic consequences. This circular describes key arthropod pests encountered in Oklahoma greenhouses and interiorscapes and provides information on IPM practices for their control. Specific pesticide recommendations are not provided but can be found in Oklahoma Cooperative Extension publications CR-6718, Management of Insects and Mites in Greenhouse Floral Crops and E-832, OSU Extension Agents’ Handbook of Insect, Plant Disease, and Weed Control. This circular replaces E-909, Commercial Greenhouse Pests.
Aphids
Aphids feed on a wide range of greenhouse crops. They are soft-bodied, pear-shaped insects with somewhat long legs and antennae. There are winged and wingless forms, varying from yellow-green to red or brown, depending on species, life stage, and reproductive stage. Regardless of species, aphids are normally 1/8 inch or less in length. Most have a pair of cornicles (structures resembling tailpipes) protruding from the upper surface of the rear end that can be seen easily using a hand lens.
Aphids feed on buds, leaves, stems, and roots by inserting their long, straw-like, piercing-sucking mouthparts (stylet's) and withdrawing plant sap. Expanding leaves from damaged buds may be curled or twisted and attacked leaves often display chloridic (yellow-white) speckles where cell contents have been removed. A secondary problem arises from sugary honeydew excreted by aphids. Leaves may appear shiny and become sticky from this material, which supports the growth of black sooty mold (a common greenhouse fungus). Aphids are also notorious for their role as vectors of many viral diseases. However, of primary concern in the greenhouse is their direct feeding damage to plant tissues and overall reduction in aesthetic quality of plants.
Aphids have great reproductive potential and can achieve damaging numbers quickly. They typically give birth to female nymphs that can mature and begin reproducing in seven to ten days in greenhouse conditions. Each aphid gives birth to between 50 and 100 nymphs during a thirty-day period, which can all occur in the absence of mating.
Winged females appear when overcrowding occurs or food supplies become depleted. When cold weather is imminent, males and females appear outdoors and mate. Eggs are laid on vegetation and often overwinter successfully in Oklahoma. Under greenhouse conditions, however, activity is continuous and overwintering can occur in any life stage.
The most common aphid in greenhouses is the green peach aphid, Myzus persicae (Figure 1). Adults are light to dark green, although pink or orange individuals occasionally occur. Three very faint lines are visible running down the upper surface of the body and a dark spot can be seen on the abdomen. Using a hand lens, a distinguishing feature can be observed: a small indentation is present at the front and top of the head. Also, the antennae are long enough to reach the cornicles, and the base of each antenna bears an inwardly projecting bump (tubercle) that is only visible with a compound microscope. Green peach aphid feeds on more than 100 plant species, including a wide variety of vegetable and ornamental plants.

Figure 1a. Green peach aphid winged adult

Figure 1b. Green peach aphid wingless adult
Key plants susceptible to aphids include: ageratum, alyssum, aster, celosia, chrysanthemum, dahlia, gerbera daisy, Easter lilies, fuchsia, hydrangea, impatiens, monarda, pansy, pepper, phlox, portulaca, primula, ranunculus, salvia, snapdragon, tomato, verbena, viola, and zinnia. Virtually all greenhouse-grown crops are susceptible in varying degrees to aphids.
Integrated Pest Management Strategies
Cultural/Mechanical Control and Monitoring
- Inspect all plants brought into the greenhouse from outdoors. Aphids can also enter the greenhouse on clothing or through open doors and vents.
- Weeds can harbor aphids, so control weeds growing under benches and outside around greenhouses to prevent recurring infestations.
- Monitor aphid populations in floral crops weekly prior to flowering. Thorough plant coverage with insecticides is more difficult to achieve when flowers are present, and some insecticides can be phytotoxic to flowers as well as other plant parts.
- Place yellow sticky cards near doors and vents to detect adult aphids. Inspect randomly selected plants for nymphs and wingless adults. Be sure to examine lower leaf surfaces, stems, and buds. Look for white, cast skins (molts) and honeydew on upper leaf surfaces.
- In some cases, high aphid mortality can be inflicted using a gentle stream of water to dislodge aphids from exposed plant surfaces.
Biological Control
- Natural enemies (predators and parasitoids) are commercially available and can be released in the greenhouse. Predators include ladybugs such as Adalia bipunctata, the predatory midge, Aphidoletes aphidimyza, and green lacewings such as Chrysopa carnea. Parasitic wasps include Aphelinus addominalis, Aphidius colemani, and A. ervi.
Chemical Control
- Widespread resistance to insecticides has been reported for many common aphid pests in the greenhouse. However, several products with novel modes of action are available that can be rotated to manage aphids, while minimizing development of resistance. Many insecticides are harmful to natural enemies, so if a biological control program is in place, choose narrow-spectrum insecticides that minimize negative effects on beneficial insects. For example, some products contain insect pathogens that specifically target the pest insect but cause only minimal harm to non-target organisms.
Fungus Gnats and Shore Flies
Fungus gnats, Bradysia spp. and Sciara spp., and shore flies, Scatella spp., were once considered minor nuisance pests in the greenhouse. Production systems that employ misting equipment or are otherwise heavily irrigated create a continuously wet soil environment favorable to fungus gnat development. Plants grown in soil rich in organic matter are also susceptible to fungus gnats. Damage is caused by the larvae, which feed on organic matter and attack bulbs, roots, and other fleshy plant parts. Larvae initially feed on root hairs and eventually tunnel into the plant stem. Delicate seedlings of many greenhouse species can be killed, with plants yellowing and often wilting rapidly before death. Adult fungus gnats and shore flies have been implicated in transmission of economically important plant pathogens including the oomycete, Pythium, and the fungi, Verticillium, Botrytis, and Fusarium. Fungal spores are typically carried from plant to plant on the bodies of adult flies, but may also be present in feces of fungus gnats. Fungus gnat larvae may also transmit Pythium, Verticillium, Fusarium, and Phoma.
Fungus gnats are delicate, dark gray to black flies with long legs and antennae (Figure 2). These insects reside on the soil surface and will fly about when pots are disturbed or watering occurs. Adults measure about 1/8 inch long and possess one pair of transparent wings. Females lay clusters of 20 to 30 eggs on moist soil surfaces, particularly soils high in organic matter. Adults live for approximately ten days, but during that period females may lay 250 to 300 eggs. Eggs hatch five to six days after oviposition (egg laying). Larvae are translucent, legless, 1/4 inch long white maggots with shiny blackheads (Figure 2). They feed for 10 to 14 days before pupating in the soil. After another five to six days, adults emerge from their pupal cases, completing their life cycle within four weeks.

Figure 2a. Darkwinged fungus gnat adult
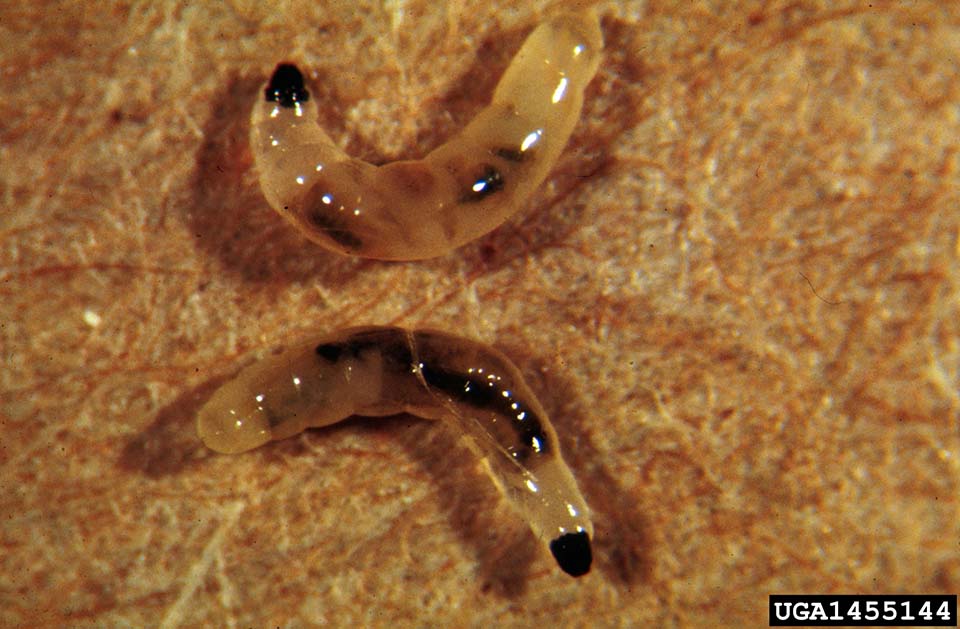
Figure 2b. Darkwinged fungus gnat larvae
Shore fly adults measure approximately 1/4 inch long, have black bodies, reddish eyes, and gray wings that contain several clear spots (Figure 3). Though often mistaken for fungus gnats, shore flies have larger, more robust bodies and relatively shorter legs and antennae. Eggs are laid in areas of algal growth and larvae hatch within four to six days following oviposition. There are three larval stages; mature larvae measure about 1/4 inch long and are opaque, yellowish brown, and lack a head capsule. The life cycle is complete in 15 to 20 days, depending on temperature. Larvae feed primarily on algae and are not known to feed on plants, but adults deposit feces on leaves, causing a reduction in aesthetic quality. Additionally, adult shore flies are known to transmit black root rot and water-mold fungus in their feces, especially under wet conditions.

Figure 3. Shore fly adult caught on a yellow sticky card. Note: clear spots on wings.
Integrated Pest Management Strategies
Cultural/Mechanical Control and Monitoring
- Avoid accumulation of water, organic matter, weeds, or debris on or under benches. Algal growth should be controlled to manage shore flies.
- Use pasteurized soil (start with clean soil and do not store or stockpile soil outdoors or uncovered).
- Inspect new plant materials carefully, especially the roots. Look for larvae, particularly in the upper inch of media in the container. It is cheaper to rogue a few plants than to control infestations later.
- To monitor adults, place yellow sticky traps horizontally and directly above the soil surface for maximum effectiveness. Fungus gnat larvae can be monitored by placing potato disks or wedges on the soil surface; wedges should be examined every two to three days for the presence of larvae.
- Avoid excessive watering and keep soil slightly dry, especially when trying to control infestations.
Biological Control
- For fungus gnat control, use the predatory mite, Hypoaspis miles, and the parasitic nematode, Steinernema feltiae. Nematodes are applied in water as a soil drench in pots or below the bench. The rove beetle, Atheta coriaria, is an effective predator of shore fly larvae.
Chemical Control
- Rotate insect growth regulators (IGR’s) with other products to control fly pests. IGR’s interfere with development and growth of immature fungus gnats and shore flies. Also, use formulations of Bacillus thuringiensis var. israelensis (Bti).
Scale Insects and Mealybugs
Greenhouse plants can serve as hosts to a wide variety of scale insects. These pests are closely related to aphids and are highly variable in shape, color, and size, although most species are less than 1/4 inch long. Scales feed by inserting their long, needle-like mouthparts into plant tissue and extracting plant sap, resulting in reduced plant vigor, stunted growth, reduced photosynthesis, and dieback. Scales are highly specialized insects that most people misdiagnose as abnormal growths or disease symptoms due to their concealment within hardened bodies or beneath dome-like, waxy covers. Common greenhouse species in Oklahoma include black scale, Saissetia oleae, hemispherical scale, Saissetia coffeae, and brown soft scale, Coccus hhesperidum (Figure 4). These species belong to a family of scales called soft scales (Coccidae). Like aphids, soft scales excrete honeydew, which can accumulate on upper leaf surfaces and support the growth of black sooty mold. Other scale pests in the greenhouse include armored scales (Diaspididae) and pit scales (Asterolecaniidae).

Figure 4a. Black scale

Figure 4b. Hemispherical scale

Figure 4c. Soft brown scale
While feeding, adults inject toxic saliva into the host plant, causing cellular damage within affected tissues. Adult females are sac-like, wingless, and typically legless. Adult males have legs and one pair of wings, but lack mouthparts and, thus, do not feed. As such, upon emergence they seek out and mate with available females, then die. In some species, males are rare or entirely lacking, and virgin females produce clones of themselves. Live young or eggs are produced under the female’s body or waxy cover. First stage nymphs are called crawlers because they have legs and move about for one or two days in search of prime feeding areas on the plant. Crawlers then insert their mouthparts into plant tissue and settle, forming their waxy cover as they feed. Most species are immobile for the remainder of their lives. Settled crawlers develop through several molts, losing their legs during the first molt. One to eight generations can occur within a year, depending on species and temperature.
Mealybugs (Pseudococcidae) are soft-bodied insects that measure about 1/8 to 1/4 inch in length. All mealybugs are covered by a white granular or cottony layer of wax. Many species possess long, waxy filaments or shorter tufts projecting outward from the body. The life cycle of most species is similar with females laying 300 to 600 eggs in cottony sacs usually located beneath the female. Following oviposition, eggs hatch in seven to ten days into tiny yellowish nymphs (crawlers). The long-tailed mealybug is slightly different because females give birth to living young. Mealybugs injure plants by sucking sap from tender foliage, flowers, stems, fruits, and roots. Heavy infestations result in stunted and distorted new growth. Like soft scales, mealybugs excrete honeydew, which can give rise to black sooty mold. Depending on species and temperature, the complete life cycle can take six weeks to two months, though mealybugs can be found in the greenhouse year round.
Many species occur on greenhouse floral crops, but perhaps the most common is the citrus mealybug, Planococcus citri (Figure 5). Citrus mealybugs have a white, powdery substance covering their bodies and white, waxy filaments projecting from the end of their abdomen. They also have shorter filaments of wax along the body margins and a faint, gray stripe running down the length of their body. This pest feeds on more than 27 families of host plants, including amaryllis, begonia, coleus, cyclamen, and dahlia.

Figure 5. Adult citrus mealybugs on Kalanchoe
Integrated Pest Management Strategies
Cultural/Mechanical Control and Monitoring
- Monitor for scales and mealybugs by closely examining stems and both sides (especially undersides) of leaves at least once per week. Look for deposits of honeydew and black sooty mold on upper leaf surfaces.
- Examine new plant material carefully for the presence of scales and mealybugs on stems and leaves.
- Pheromone traps can be used to capture male citrus mealybugs for monitoring the presence of mealybugs in greenhouses.
Biological Control
- Soft scales are controlled with parasitic wasps released into the greenhouse. Many wasp species specialize on one or several scale insects, so be sure to properly match the biological control agent with its scale host. For instance, Metaphycus luteola is very effective at controlling brown soft scale, but M. helvolus is better suited to controlling hemispherical scale.
- Various parasitic wasps for controlling mealybugs are available commercially. Citrus mealybug is effectively controlled using Leptomastix dactylopii in conjunction with the ladybug predator, Cryptolaemus montrouzieri, known as mealybug destroyer.
Chemical Control
- Most scale species, especially soft scales, and mealybugs are effectively controlled with systemic insecticides. Armored scales can be controlled or at least suppressed with certain systemic insecticides, but horticultural oil can be used very effectively against armored scales.
- Commercially available insecticidal soaps are also an effective option.
- Minor mealybug infestations are often controlled with a 50/50 mixture of common rubbing alcohol and water.
Whiteflies
Whiteflies are not true flies, but are close relatives of aphids and scales. They measure 1/16 inch long, have four wings, and are orange underneath. They resemble small white moths due to the white, waxy powder on top of their bodies (Figure 6). Whiteflies reside on the underside of leaves and will fly readily when disturbed. During heavy infestations, a “white cloud” appears during watering or when plants are disturbed. These insects feed through piercing-sucking mouthparts, sometimes causing stippling (small, discolored spots) on leaves. Plants eventually lose vigor as essential nutrients are withdrawn with the plant sap. Like their close relatives, whiteflies excrete honeydew, supporting the growth of black sooty mold on plant surfaces. Whiteflies are problematic for several reasons: they have high reproductive rates, rapidly develop resistance to insecticides, some can transmit plant viruses, and they attack a wide range of major ornamental crops. Poinsettia, chrysanthemum, fuchsia, many bedding plants, and tomatoes are particularly susceptible to whiteflies.

Figure 6. Adult and larvae of greenhouse whitefly
Despite the fact that they are tropical insects, whiteflies are found in greenhouses year round. They spread from greenhouse to greenhouse via transport of host plants and by attaching to workers’ clothing. Yellow clothing should not be worn by workers as this color is particularly alluring to whiteflies. Several species are of economic importance to greenhouse production, including greenhouse whitefly, Trialeurodes vaporariorum, sweetpotato whitefly, Bemisia tabaci biotype B (also known as silverleaf whitefly, Bemisia argentifolii), and sweetpotato whitefly, Bemisia tabaci biotype Q. Biotypes B and Q of sweetpotato whitefly cannot be distinguished based on physical traits, so only DNA tests can confirm their true identity. It is important to make the correct determination because biotype Q is resistant to several classes of insecticides. Contact your county Extension office to inquire about how to determine the predominant biotype present in your greenhouse.
Female whiteflies usually deposit eggs in a circular pattern and most frequently on the underside of younger, upper foliage. Adult females may lay as many as 150 to 250 eggs in their lifetime. Eggs are tiny, spindle shaped, and attached to the leaf surface by a thin stalk. Eggs appear creamy when laid but eventually darken. Newly hatched nymphs emerge from the eggs in five to ten days and seek feeding sites. After inserting their mouthparts into leaf tissue, they remain stationary for three weeks, during which three molts occur. All nymphal stages are flat, transparent to yellow-green, and appear scale-like. At the end of this period, the whitefly transforms into a non-feeding, yellowish-green “pseudopupa” with two conspicuous eyes. About one week later, the winged adult emerges and begins laying eggs within a week. The entire life cycle is complete within four to five weeks, depending on temperature.
Sweetpotato whitefly holds its wings closer to the body and appears smaller than greenhouse whitefly. Damage is similar to that of greenhouse whitefly, but sweet potato whitefly can reproduce more aggressively, particularly because insecticide resistance is acquired rapidly with this species. Poinsettias are extremely susceptible to sweet potato whitefly. Because whitefly populations can increase quickly, it is essential to monitor their populations early in the production schedule. Early vigilance in poinsettia crops is also important, because plants are susceptible to pesticide damage once they begin showing color.
Integrated Pest Management Strategies
Cultural/Mechanical Control and Monitoring
- Monitor for sessile stages of whitefly and their eggs by closely examining undersides of leaves, especially early in the production cycle. Look for deposits of honeydew and black sooty mold on upper leaf surfaces.
- Weeds can be an important source of whiteflies, so control weeds growing under benches to prevent recurring whitefly problems.
Biological Control
- Several parasitic wasps are commercially available for whitefly control. Encarsia formosa attacks greenhouse whitefly larvae and Eretmocerus eremicus parasitizes both greenhouse,and silverleaf (current name for sweet potato) whitefly. Eretmocerus mundus specifically controls silverleaf whitefly and can survive under a variety of climatic conditions. This wasp can be introduced into the greenhouse at any time, including early in the season. The predatory mite, Amblyseius swirskii, feeds on whitefly eggs and larvae as well as thrips. This predator performs best when both prey items are available. Delphastus catalinae, a small black lady beetle, is also commercially obtainable.
Chemical Control
- Systemic and some contact insecticides have provided effective control of whiteflies, but be sure to rotate among chemical classes to prevent the development of resistance. Contact insecticides should be directed at the underside of leaves and applied when first- and second-instar whitefly larvae are present.
Thrips
Most thrips species measure no more than 1/8 inch long, have slender bodies, and have two pairs of stalk-like wings fringed with long hairs (Figure 7). Populations often proliferate on weeds growing around greenhouses during summer months and may be carried in by air currents through open vents or doors (especially during wheat harvest in Oklahoma). Thrips attack a wide range of crops and can be found in buds, on flower petals, and in axils of leaves. While adults can be seen without a hand lens, they usually hide in buds or flowers. Thrips can be detected by thorough visual inspection of plants and by tapping foliage, buds, or flowers over white paper. Adults can be yellow, brown, tan, or black, depending on species.

Figure 7. Onion thrips (left) and western flower thrips (right).
Using their saw-like ovipositor, adult females lay their eggs in leaves and other plant tissue. In two to eight days, eggs hatch, and larvae immediately begin to feed. After feeding for ten to fourteen days, larvae burrow into the soil and pupate. Adults emerge about four days later. The life cycle is complete in as little as two weeks, though cooler temperatures may delay completion of the life cycle up to a month.
Thrips have rasping mouthparts that scrape tender leaves and petals, creating a wound allowing them to suck exuding plant sap. Thrips injury appears as white or silver streaks on affected plant parts, which turn tan or brown as cells die. In addition, thrips feeding on newly expanding leaves can distort tissue severely, resulting in cupping (curled leaf margins) or irregularly shaped leaves, buds, and flowers. A sign of an infestation is the presence of green to black fecal droppings on leaves and petals.
Adults and larvae transmit two important tospoviruses, tomato spotted wilt virus (TSWV) and impatiens necrotic spot virus (INSV). The predominant vector of these important plant diseases is western flower thrips, Frankliniella occidentalis. Male western flower thrips are pale yellow and have a narrow abdomen; whereas females are larger, yellow to dark brown, and have a more rounded abdomen. Each female may live up to six weeks, and may produce 150 to 250 eggs during her lifetime. Larvae develop through two instars and are yellow.
Thrips attack at least 300 species of plants. New Guinea impatiens, chrysanthemums, and gloxinias are especially susceptible to tospoviruses, and symptoms vary with host plant from bulls-eye patterns on foliage to brown sap oozing from lesions on stems. Plants infected with TSWV or INSV must be destroyed because the diseases are incurable. Therefore, controlling thrips is important in preventing transmission of these economically important diseases as well as reduced aesthetic quality due to feeding. Plants showing symptoms of disease can be submitted to the Plant Disease and Insect Diagnostic Lab at Oklahoma State University for evaluation. Instructions for preparation and submission of samples can be found at Plant Disease and Insect Diag Lab (PDIDL).
Integrated Pest Management Strategies
Cultural/Mechanical Control and Monitoring
- Know your source of plants. Do not introduce new plants that may have the virus into a greenhouse containing healthy plants. Thrips can quickly spread the virus to epidemic levels.
- Eliminate sources of thrips by removing weeds growing under benches and outside around greenhouses.
- Exclude thrips from the greenhouse by installing vent/fan screens and double doors.
- Monitor for adult thrips with blue or yellow sticky cards placed just above the plant canopy and check cards at least once per week. If adults are detected, closely examine plants around the sticky card for thrips and plant damage.
- Thrips can also be detected by tapping foliage, buds, or flowers over white paper.
Biological Control
- Predatory mites such as Hypoaspis miles, Amblyseius cucumeris, and A. swirskii, and the minute pirate bug, Orius insidiosus, are commercially available for release against thrips. Amblyseius mites should be released before thrips populations build up to damaging levels. The soil-inhabiting mite, Hypoaspis miles, is available for release against thrips pupae, root-infesting aphids, and fungus gnat larvae. The parasitic nematode, Steinernema feltiae, is applied in water as a soil drench to control thrips pupae.
Chemical Control
- Thrips are normally controlled with contact insecticides, but acceptable levels of control are often difficult to achieve because thrips inhabiting flowers are often well protected from sprays. Systemic insecticides will not work against thrips feeding on flower buds or petals because the active ingredients cannot be transported to those tissues.
- If a biological control program is in place, choose selective insecticides such as IGR’s or products containing the insect-killing fungus, Beauveria bassiana, to minimize harmful effects on natural enemies, especially predatory mites.
Mites
Mites are arachnids, not insects, and thus are more closely related to scorpions and spiders. Unlike insects, adult mites have four pairs of legs regardless of the species. However, immature mites can have three pairs of legs early in their life cycle, so be careful when trying to diagnose a pest problem.
Probably the most common mite in the greenhouse is the two-spotted spider mite, Tetranychus urticae. This mite measures about 1/50 inch long, requiring at least a 10X hand lens to aid in its identification. Two-spotted spider mites may be yellow or green with two, and occasionally four, dark spots on their bodies (Figure 8). This mite feeds by piercing leaf tissue with its thin, whip-like mouthparts and sucking the plant sap exuded at the wound site. Feeding causes chlorotic stippling of leaves, giving the appearance of fine, yellowish to white speckling on the foliage. Stippling is distinctive and is readily recognized by experienced growers. Spider mites are most likely to be found on the underside of leaves and in the flowers. As populations increase, spider mites often spin webbing over foliage and flowers (Figure 8).

Figure 8a. Adult two-spotted spider mites

Figure 8b. Gorse spider mite web
Duration of the life cycle varies greatly with temperature in the greenhouse. But under hot, dry conditions, it may take as little as seven days for mites to develop from egg to adult. At 70°F the life cycle is twenty days but only half that when temperatures are at 80°F. High temperatures and low relative humidity favor mite development. In two to five days, eggs hatch into six-legged larvae that feed for a short time. Larvae then develop into eight-legged nymphs that pass through two stages, the protonymph and deutonymph. Between each stage, larvae and nymphs enter an inactive resting stage that lasts a short time. The eight-legged adult finally emerges from the last resting stage. Because each female may deposit 100 eggs or more during her lifetime, there is a high potential for rapid population growth.
Cyclamen mites, Phytonemus pallidus, are only 1/100 inch long at maturity. These mites cannot be seen without high magnification and are translucent with a brownish tinge. Cyclamen mites attack a wide range of plants, feeding on buds and adjacent immature leaves. They attack growing points of plant tissue, which causes twisting and stunting of affected tissue, and new growth may appear purplish. Damaged leaflets curl from outside inward because feeding causes small depressions to form in young leaves. Damaged flower buds may grow into distorted flowers or may not open at all.
High humidity (80 percent or more) with a cool temperature (60°F or less) favors their development. The life cycle from egg to adult can occur in only two weeks, with the female living up to four weeks and laying up to 100 eggs in her lifetime.
Integrated Pest Management Strategies
Cultural/Mechanical Control and Monitoring
- Monitor for mites by examining plants closely for stippling, webbing, and/or herbicide-like damage. Be especially vigilant when ambient conditions are conducive to the development of spider mites (hot and dry) and cyclamen mites (cool and wet).
- Like thrips, spider mites can be detected by careful visual inspection and by tapping foliage, buds, or flowers over white paper.
- Highly susceptible plants can be used as indicator plants to a developing cyclamen mite problem. For example, African violets will not bloom, followed by the development of hairy, stunted leaves. Impatiens and petunias will exhibit distorted terminal growth.
Biological Control
- Amblyseius californicus, Phytoseiulus persimilis, and Neoseiulus fallacis are predatory mites that can be used to control two-spotted spider mites. A predatory midge, Feltiella acarisuga, is also available for spider mite control. While relatively new, a small lady beetle, Stethorus punctillum, is now available and has the advantage of flying to its prey.
Chemical Control
- Several miticides are available for mite control, but be sure to check which life stages are controlled by each product. Resting stages tend to be more tolerant of miticides, but some newer products can kill eggs as well as other life stages.
- Repeated applications of contact insecticides may be necessary when mites are abundant. However, care must be taken in choosing a product when biological control programs are in place.
- During hot Oklahoma summers, it may be necessary to schedule pesticide applications as often as two days apart or to be as aggressive as the label permits.
- Cyclamen mites can be killed by immersing infested plant material in hot water (110°F) for fifteen minutes. Different plants should be tested for sensitivity to hot water before attempting this treatment.
Leaf Miners
A variety of insects mine leaves in nature, but several species from a family of flies (Agromyzidae) are the primary leaf miner pests in greenhouses. Adult leaf miners are stocky flies that measure about 1/12 inch long (Figure 9). Using their ovipositor, females puncture the leaf surface and insert tiny eggs within the leaf. Each female can lay up to 100 eggs in her two- to three-week life span. These puncture wounds turn white shortly after oviposition, giving the leaf a speckled appearance following multiple attacks. In five to seven days, eggs hatch into white maggots that grow to 1/10 inch long at maturity. Larvae tunnel within leaves for about two weeks, then become pupae. Depending on species, pupation takes place in or on the soil, in leaf mines, or on leaf surfaces. About two weeks later, the adult fly emerges from the pupa, repeating the life cycle. Leaf miners can survive transport of cuttings in the egg, larval, or pupal stage, so infestations may result from shipment of infested material.

Figure 9. Adult chrysanthemum leaf miner.
Tunneling maggots feed just under the outer cell layer of leaves, causing unsightly runs or mines as they feed. Damage causes reduced aesthetic quality, but tunneling usually does not kill the plant. Problematic greenhouse species include the serpentine leaf miner, Liriomyza trifolii, and the chrysanthemum leaf miner, Phytomyza atricornis. Adult serpentine leaf miners have a black body with yellow markings, a yellow head, and brown eyes. As its name suggests, mines created by this species are serpentine in appearance. Adult chrysanthemum leaf miners are larger and grey to black with many hairs on their body. Their mines can appear blotchy as well as serpentine and tend to be found near the leaf mid-vein.
Integrated Pest Management Strategies
Cultural/Mechanical Control and Monitoring
- Regularly monitor susceptible plants, including aster, chrysanthemum, dahlia, and gerbera daisy. Remove mined leaves before adult flies emerge and be sure to destroy infested foliage.
Biological Control
- Three parasitic wasps, Dacnusa sibirica, Opius pallipes, and Diglyphus isaea, have been used successfully for leaf miner control.
Chemical Control
- Serpentine leaf miner is highly resistant to organophosphate, carbamate, and pyrethroid insecticides. In general, some systemic insecticides and IGR’s can effectively control leaf miners.
Caterpillars
Larvae of several species of moths (caterpillars) can attack greenhouse crops. Caterpillars vary in appearance, but all have chewing mouthparts, three pairs of segmented “true” legs on the first three body segments behind the head, and five or fewer pairs of fleshy, unsegmented, abdominal prolegs (Figure 10). These prolegs have tiny hook-like structures called crochets at the tips that allow the caterpillar to grip stems, petioles, and foliage.

Figure 10. Beet armyworm caterpillar. Note the three pairs of true legs near the head and five pairs of fleshy, abdominal prolegs.
Feeding damage occurs on leaves when the caterpillar chews holes or “skeletonizes” leaf tissue (feeding only on tissue between veins). A severe infestation of caterpillars can quickly defoliate entire plants. Injury may not be noticed when caterpillars are young, but damage becomes increasingly evident as these pests develop and grow. Caterpillars are particularly troublesome during the summer months when moths are active outdoors. Most moths are nocturnal and attracted to lights, entering a greenhouse through open doors or vents. Mated female moths then proceed to lay eggs on greenhouse crops. Beet armyworm, corn earworm, European corn borer, and various cutworms, leaf tiers, and leaf rollers are a few of many caterpillars that can occasionally damage tender plants.
Control is similar for most caterpillar pests, with stomach poisons being commonly used. Control with Bacillus thuringiensis is often effective, but efficacy is greatest against young caterpillars. Repeat applications of this bacterium are necessary to keep new growth surfaces covered as plants grow. If caterpillars enter stems or buds, they become very difficult to control.
Integrated Pest Management Tactics
Cultural/Mechanical Control and Monitoring
- Exclude moths by installing vent screens and keeping doors closed at night.
- Regularly monitor plants for symptoms of feeding damage and signs of caterpillars such as egg masses, cast skins, frass (solid waste), or the insects themselves.
- Remove and destroy any caterpillars or foliage containing egg masses.
Biological Control
- Parasitic wasps in the genus Trichogramma are commercially available and will attack moth egg masses.
Chemical Control
- Reduced-risk “biopesticides” contain insect-killing pathogens that can be highly effective against caterpillar pests and are safe for greenhouse workers and natural enemies. Choose insecticides containing the bacterium, Bacillus thuringiensis (Bt) var. kurstaki, the fungus, Beauveria bassiana, or the active ingredient, spinosad. These products are most effective against young caterpillars. Other insecticides can be used to control older larvae and adults.
Springtails
Springtails are very small, white, gray, black, brown, or purple insects that lack wings (Figure 11). A specialized structure beneath the tip of the abdomen enables them to jump an inch or more into the air. Springtails prefer dark, damp areas high in organic matter. They feed primarily on fungi, algae, and decaying organic matter in growing media. However, feeding may occur on tender roots of some crops. Damage may occur to seedlings, but springtails rarely cause enough harm to be considered major pests. When large populations occur, they are easily found on the soil surface or can be seen “springing” about during watering or moving a crop. Springtails may be spread in unpasteurized potting soil, transported on clothing, via plant contact, or simply migrating indoors from nearby habitats.
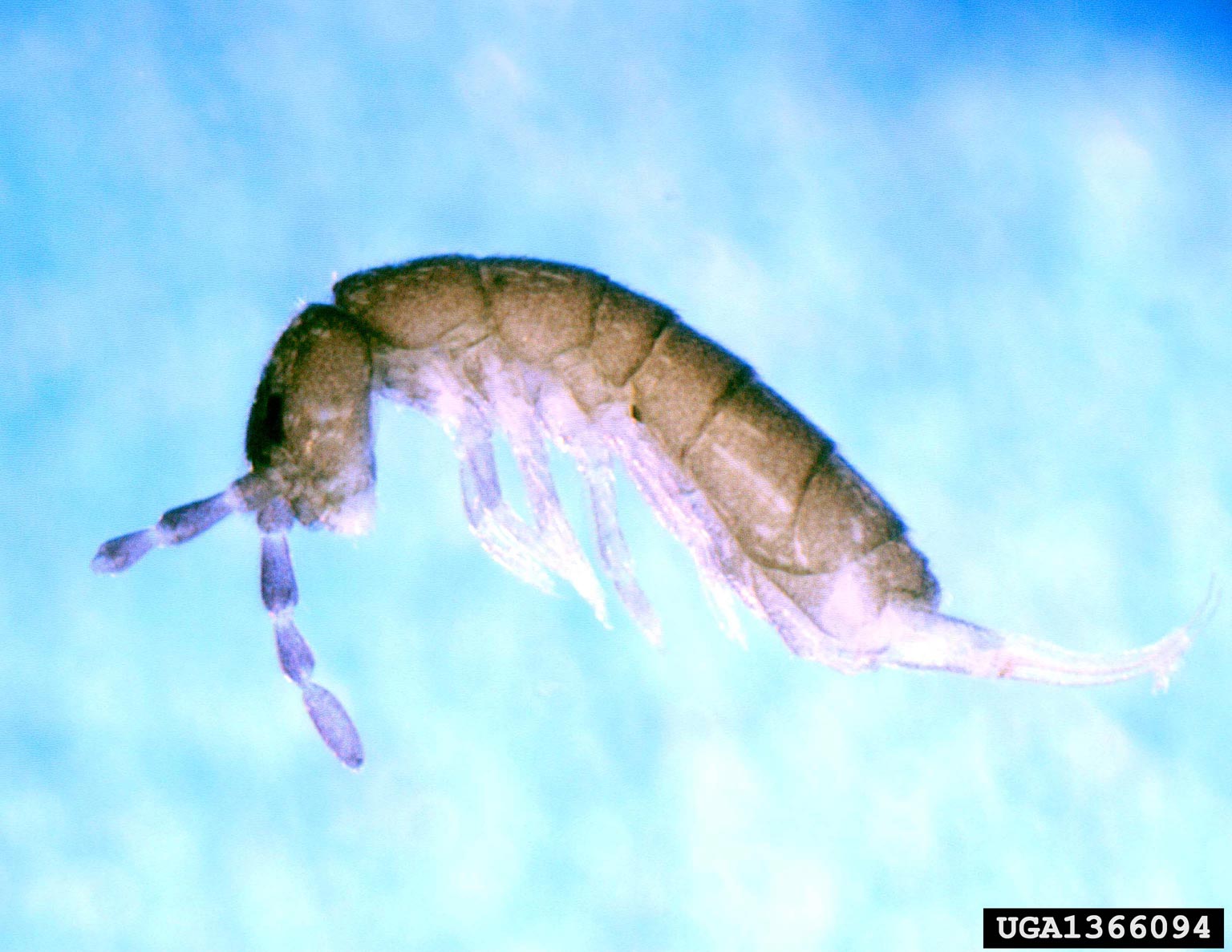
Figure 11. Springtail
Integrated Pest Management Tactics
Sanitation and soil pasteurization are most helpful in preventing or controlling springtails. The predatory mite, Hypoaspis miles, will feed on springtails as well as other soil-dwelling pests.
Slugs and Snails
Slugs and snails are mollusks rather than insects and are related to clams and oysters. Whereas snails have a hard shell, slugs lack a shell (Figure 12). Both vary greatly in size, ranging from one-half to four inches in length. Clusters of 20 to 100 eggs are laid by slugs or snails in moist crevices in the soil or in containers. Eggs hatch in ten days or less, with maturity occurring within three months to one year. Using their rasping mouthparts, slugs and snails feed at night on tender seedlings and leaves. Feeding damage appears as irregularly shaped holes with smooth edges, and may be confused with injury caused by caterpillars. Seedling plants can be completely consumed. During the day, slugs and snails hide beneath pots, benches, plant debris, or other concealed locations. Both prefer moist areas, so greenhouses can be ideal environments for their growth and development. Slugs and snails exude a slippery liquid from a muscular “foot” as they travel. When this substance dries, a shiny track is left behind. Besides feeding damage, this track is usually the best way to detect the presence of these pests.

Figure 12. Gray garden slug
Integrated Pest Management Tactics
Cultural/Mechanical Control and Monitoring
- Practice good sanitation and keep problematic areas as dry as possible.
- Inspect incoming plants and pots for slugs and snails.
- Eliminate sources of slugs and snails by removing weeds growing both inside and outside around greenhouses.
- Slugs avoid copper, so wrap copper tape around bench legs or install copper flashing around raised beds.
Biological Control
- No predators or parasites are commercially available in the U.S., although research is ongoing in this area.
Chemical Control
- Chemical baits are commercially available for slug control. Baits are usually formulated in bran and must be eaten by slugs to be effective. Place baits in the evening when slugs are most active and irrigate prior to bait placement to draw out slugs, leading to more frequent contact with the bait.
- Rotate among active ingredients to counter avoidance and resistance to baits.
General IPM Practices for Greenhouses and Interiorscapes
- Establish and maintain a thorough record-keeping system for all pest management practices.
- Become familiar with each crop’s potential pest problems.
- Conduct a pre-crop inspection of production areas for potential pest problems.
- Always inspect incoming plants for pests immediately upon arrival. Reject the shipment rather than treating or accepting “slight” infestations.
- Inspect crops for signs and symptoms of pests on a regular basis.
- Choose appropriate control methods carefully and know when to apply them.
- Discard and destroy severely infested plants.
- Physically exclude or confine pests.
- Always pasteurize media that has soil as a component.
- Consider using biological controls whenever possible.
Oklahoma Pest Control Supply Companies
-
American Plant Products and Services, Inc.
9200 Northwest 10th Street
Oklahoma City, OK 73127
405-787-4833
-
Winfield Solutions regional locations/offices:
1819 NW 5th St
Oklahoma City, OK 73106
405-232-2493
11391 East Tecumseh St.
Tulsa, OK 74116
918-234-0560
801 North 54th St.
Enid, OK 73701
580-234-8004
-
Trécé, Inc.
7569 Highway 28 West
Adair, OK 74330
918-785-3061
Other Companies Outside Oklahoma:
-
Alpha Scents, Inc.
1089 Willamette Falls Drive
West Linn, OR 97068
(503) 342-8611
Alpha Scents, Inc.
-
BioWorks
100 Rawson Road
Suite 205
Victor, NY 14564
800-877-9443
-
IPM Laboratories, Inc.
Locke, NY
315-497-2063
-
Hydro-Gardens
8765 Vollmer Rd
Colorado Springs, CO 80908
888-693-0578
Eric J. Rebek
Extension Entomologist/ Ornamental and Turfgrass Specialist
Michael A. Schnelle
Extension Ornamental/ Floriculture Specialist
